Input interpretation

period 5 elements | atomic diameter
Summary
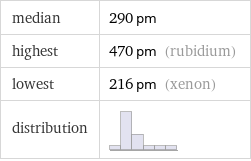
median | 290 pm highest | 470 pm (rubidium) lowest | 216 pm (xenon) distribution |
Units

Plot
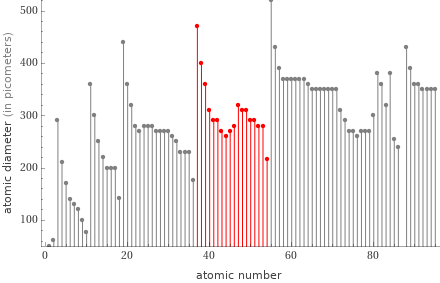
Plot
Periodic table location

Periodic table location
Atomic radii properties
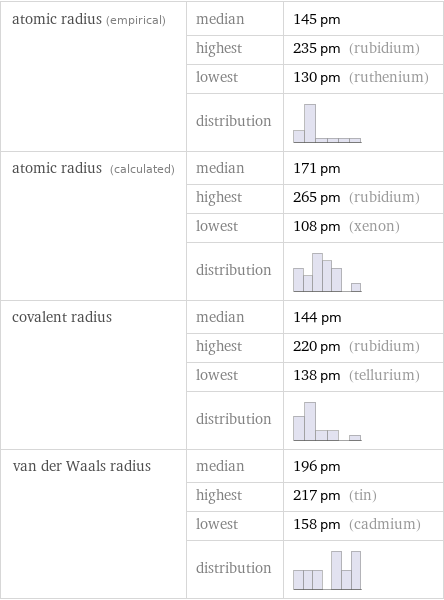
atomic radius (empirical) | median | 145 pm | highest | 235 pm (rubidium) | lowest | 130 pm (ruthenium) | distribution | atomic radius (calculated) | median | 171 pm | highest | 265 pm (rubidium) | lowest | 108 pm (xenon) | distribution | covalent radius | median | 144 pm | highest | 220 pm (rubidium) | lowest | 138 pm (tellurium) | distribution | van der Waals radius | median | 196 pm | highest | 217 pm (tin) | lowest | 158 pm (cadmium) | distribution |
Units

Distribution plots
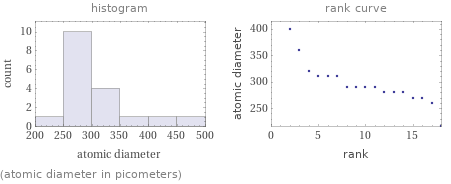
(atomic diameter in picometers)
Atomic diameter rankings
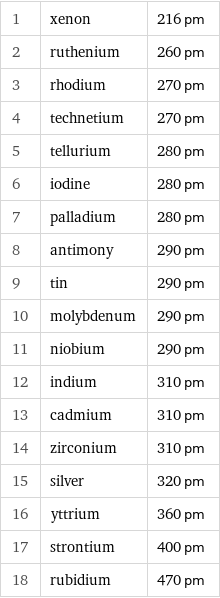
1 | xenon | 216 pm 2 | ruthenium | 260 pm 3 | rhodium | 270 pm 4 | technetium | 270 pm 5 | tellurium | 280 pm 6 | iodine | 280 pm 7 | palladium | 280 pm 8 | antimony | 290 pm 9 | tin | 290 pm 10 | molybdenum | 290 pm 11 | niobium | 290 pm 12 | indium | 310 pm 13 | cadmium | 310 pm 14 | zirconium | 310 pm 15 | silver | 320 pm 16 | yttrium | 360 pm 17 | strontium | 400 pm 18 | rubidium | 470 pm
Unit conversions for median atomic diameter 290 pm
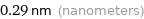
0.29 nm (nanometers)
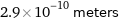
2.9×10^-10 meters
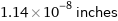
1.14×10^-8 inches
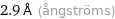
2.9 Å (ångströms)
Comparisons for median atomic diameter 290 pm
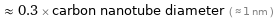
≈ 0.3 × carbon nanotube diameter ( ≈ 1 nm )
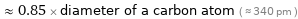
≈ 0.85 × diameter of a carbon atom ( ≈ 340 pm )
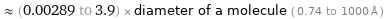
≈ (0.00289 to 3.9) × diameter of a molecule ( 0.74 to 1000 Å )