Input interpretation
dibromoneopentyl glycol
Chemical names and formulas

formula | HOCH_2C(CH_2Br)_2CH_2OH Hill formula | C_5H_10Br_2O_2 name | dibromoneopentyl glycol IUPAC name | 2, 2-bis(bromomethyl)propane-1, 3-diol alternate names | 1, 3-dibromo-2, 2-dihydroxymethylpropane | 1, 3-propanediol, 2, 2-bis(bromomethyl) | 1, 3-propanediol, 2, 2-bis(bromomethyl)- | 2, 2-bis(bromomethyl)-1, 3-propanediol | 2, 2-bis(bromomethyl)propane-1, 3-diol | pentaerythritol dibromide mass fractions | Br (bromine) 61% | C (carbon) 22.9% | H (hydrogen) 3.85% | O (oxygen) 12.2%
Lewis structure

Draw the Lewis structure of dibromoneopentyl glycol. Start by drawing the overall structure of the molecule: Count the total valence electrons of the bromine (n_Br, val = 7), carbon (n_C, val = 4), hydrogen (n_H, val = 1), and oxygen (n_O, val = 6) atoms: 2 n_Br, val + 5 n_C, val + 10 n_H, val + 2 n_O, val = 56 Calculate the number of electrons needed to completely fill the valence shells for bromine (n_Br, full = 8), carbon (n_C, full = 8), hydrogen (n_H, full = 2), and oxygen (n_O, full = 8): 2 n_Br, full + 5 n_C, full + 10 n_H, full + 2 n_O, full = 92 Subtracting these two numbers shows that 92 - 56 = 36 bonding electrons are needed. Each bond has two electrons, so the above diagram has all the necessary bonds. There are 18 bonds and hence 36 bonding electrons in the diagram. Lastly, fill in the remaining unbonded electrons on each atom. In total, there remain 56 - 36 = 20 electrons left to draw: Answer: | |
3D structure

3D structure
Basic properties
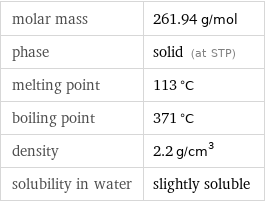
molar mass | 261.94 g/mol phase | solid (at STP) melting point | 113 °C boiling point | 371 °C density | 2.2 g/cm^3 solubility in water | slightly soluble
Units

Solid properties (at STP)

density | 2.2 g/cm^3 vapor pressure | 9.998 mmHg
Units

Thermodynamic properties

molar heat of vaporization | 71.5 kJ/mol specific heat of vaporization | 0.273 kJ/g (at STP)
Chemical identifiers

CAS number | 3296-90-0 PubChem CID number | 18692 PubChem SID number | 24858219 SMILES identifier | C(C(CO)(CBr)CBr)O InChI identifier | InChI=1/C5H10Br2O2/c6-1-5(2-7, 3-8)4-9/h8-9H, 1-4H2 RTECS number | TY3195500 MDL number | MFCD00004688
Safety properties

flash point | 178 °C
Toxicity properties

RTECS classes | tumorigen | mutagen | reproductive effector