Input interpretation
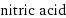
nitric acid
Chemical names and formulas

formula | HNO_3 name | nitric acid alternate names | anhydrous nitric acid | azotic acid | hydrogen nitrate | nital | spirit of nitre mass fractions | H (hydrogen) 1.6% | N (nitrogen) 22.2% | O (oxygen) 76.2%
Lewis structure

Draw the Lewis structure of nitric acid. Start by drawing the overall structure of the molecule, ignoring potential double and triple bonds: Count the total valence electrons of the hydrogen (n_H, val = 1), nitrogen (n_N, val = 5), and oxygen (n_O, val = 6) atoms: n_H, val + n_N, val + 3 n_O, val = 24 Calculate the number of electrons needed to completely fill the valence shells for hydrogen (n_H, full = 2), nitrogen (n_N, full = 8), and oxygen (n_O, full = 8): n_H, full + n_N, full + 3 n_O, full = 34 Subtracting these two numbers shows that 34 - 24 = 10 bonding electrons are needed. Each bond has two electrons, so in addition to the 4 bonds already present in the diagram add 1 bond. To minimize formal charge oxygen wants 2 bonds. Identify the atoms that want additional bonds and the number of electrons remaining on each atom: To fully fill its valence shell, nitrogen will donate one of its electrons, allowing it to form four bonds (the maximum number an element on period 2 can form). Fill in the 1 bond by pairing electrons between adjacent highlighted atoms, noting the formal charges of the atoms. Double bonding nitrogen to the other highlighted oxygen atom would result in an equivalent molecule: Answer: | |
3D structure

3D structure
Basic properties
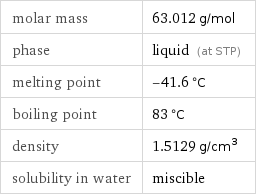
molar mass | 63.012 g/mol phase | liquid (at STP) melting point | -41.6 °C boiling point | 83 °C density | 1.5129 g/cm^3 solubility in water | miscible
Units

Liquid properties (at STP)
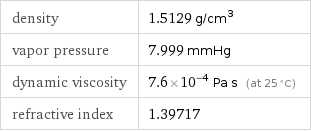
density | 1.5129 g/cm^3 vapor pressure | 7.999 mmHg dynamic viscosity | 7.6×10^-4 Pa s (at 25 °C) refractive index | 1.39717
Units
Thermodynamic properties
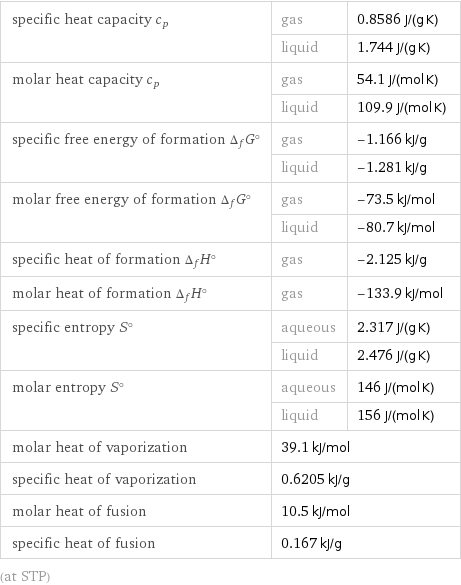
specific heat capacity c_p | gas | 0.8586 J/(g K) | liquid | 1.744 J/(g K) molar heat capacity c_p | gas | 54.1 J/(mol K) | liquid | 109.9 J/(mol K) specific free energy of formation Δ_fG° | gas | -1.166 kJ/g | liquid | -1.281 kJ/g molar free energy of formation Δ_fG° | gas | -73.5 kJ/mol | liquid | -80.7 kJ/mol specific heat of formation Δ_fH° | gas | -2.125 kJ/g molar heat of formation Δ_fH° | gas | -133.9 kJ/mol specific entropy S° | aqueous | 2.317 J/(g K) | liquid | 2.476 J/(g K) molar entropy S° | aqueous | 146 J/(mol K) | liquid | 156 J/(mol K) molar heat of vaporization | 39.1 kJ/mol | specific heat of vaporization | 0.6205 kJ/g | molar heat of fusion | 10.5 kJ/mol | specific heat of fusion | 0.167 kJ/g | (at STP)
Chemical identifiers
(O)[O-] InChI identifier | InChI=1/HNO3/c2-1(3)4/h(H, 2, 3, 4)/f/h2H MDL number | MFCD00011349](../image_source/bbab7829cdacfe43f379f5972ca0f783.png)
CAS number | 7697-37-2 PubChem CID number | 944 PubChem SID number | 24853516 SMILES identifier | [N+](=O)(O)[O-] InChI identifier | InChI=1/HNO3/c2-1(3)4/h(H, 2, 3, 4)/f/h2H MDL number | MFCD00011349
NFPA label

NFPA label
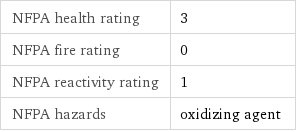
NFPA health rating | 3 NFPA fire rating | 0 NFPA reactivity rating | 1 NFPA hazards | oxidizing agent
Toxicity properties

short-term exposure limit | 10 mg/m^3 threshold limit value | 2 ppmv

long-term exposure limit | 5 mg/m^3 (over 8 hours) RTECS classes | other
Ion equivalents

H^+ (hydrogen cation) | 1 (NO_3)^- (nitrate anion) | 1