Input interpretation

indium (chemical element) | rhodium (chemical element) | palladium (chemical element)
Periodic table location
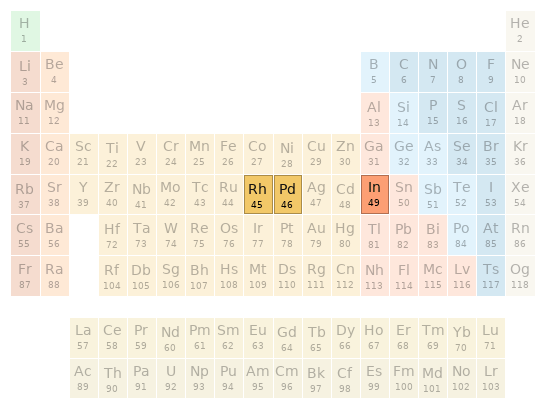
Periodic table location
Images

Images
Basic elemental properties
![| indium | rhodium | palladium atomic symbol | In | Rh | Pd atomic number | 49 | 45 | 46 short electronic configuration | [Kr]5s^24d^105p^1 | [Kr]5s^14d^8 | [Kr]4d^10 Aufbau diagram | 5p 4d 5s | 4d 5s | 4d block | p | d | d group | 13 | 9 | 10 period | 5 | 5 | 5 atomic mass | 114.818 u | 102.9055 u | 106.42 u half-life | (stable) | (stable) | (stable)](../image_source/43ca92ea6b4d4e66c992c8463b599f2d.png)
| indium | rhodium | palladium atomic symbol | In | Rh | Pd atomic number | 49 | 45 | 46 short electronic configuration | [Kr]5s^24d^105p^1 | [Kr]5s^14d^8 | [Kr]4d^10 Aufbau diagram | 5p 4d 5s | 4d 5s | 4d block | p | d | d group | 13 | 9 | 10 period | 5 | 5 | 5 atomic mass | 114.818 u | 102.9055 u | 106.42 u half-life | (stable) | (stable) | (stable)
Thermodynamic properties
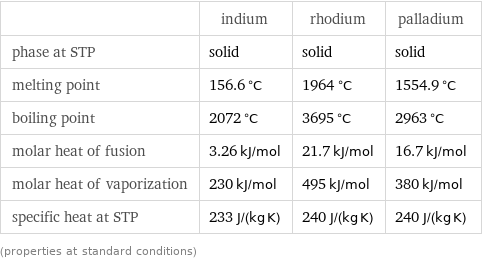
| indium | rhodium | palladium phase at STP | solid | solid | solid melting point | 156.6 °C | 1964 °C | 1554.9 °C boiling point | 2072 °C | 3695 °C | 2963 °C molar heat of fusion | 3.26 kJ/mol | 21.7 kJ/mol | 16.7 kJ/mol molar heat of vaporization | 230 kJ/mol | 495 kJ/mol | 380 kJ/mol specific heat at STP | 233 J/(kg K) | 240 J/(kg K) | 240 J/(kg K) (properties at standard conditions)
Material properties
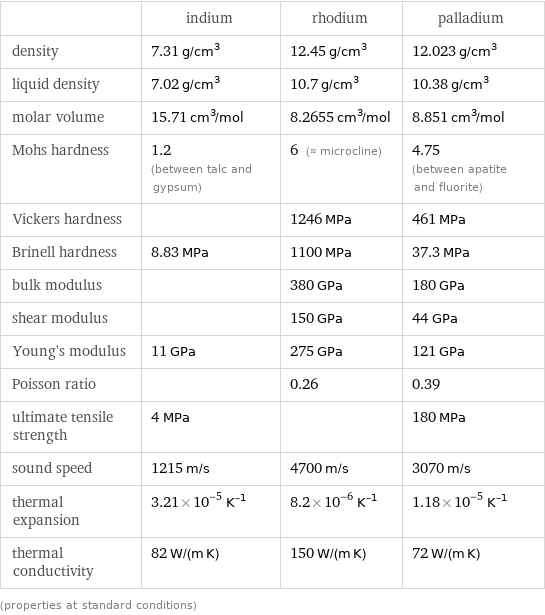
| indium | rhodium | palladium density | 7.31 g/cm^3 | 12.45 g/cm^3 | 12.023 g/cm^3 liquid density | 7.02 g/cm^3 | 10.7 g/cm^3 | 10.38 g/cm^3 molar volume | 15.71 cm^3/mol | 8.2655 cm^3/mol | 8.851 cm^3/mol Mohs hardness | 1.2 (between talc and gypsum) | 6 (≈ microcline) | 4.75 (between apatite and fluorite) Vickers hardness | | 1246 MPa | 461 MPa Brinell hardness | 8.83 MPa | 1100 MPa | 37.3 MPa bulk modulus | | 380 GPa | 180 GPa shear modulus | | 150 GPa | 44 GPa Young's modulus | 11 GPa | 275 GPa | 121 GPa Poisson ratio | | 0.26 | 0.39 ultimate tensile strength | 4 MPa | | 180 MPa sound speed | 1215 m/s | 4700 m/s | 3070 m/s thermal expansion | 3.21×10^-5 K^(-1) | 8.2×10^-6 K^(-1) | 1.18×10^-5 K^(-1) thermal conductivity | 82 W/(m K) | 150 W/(m K) | 72 W/(m K) (properties at standard conditions)
Electromagnetic properties
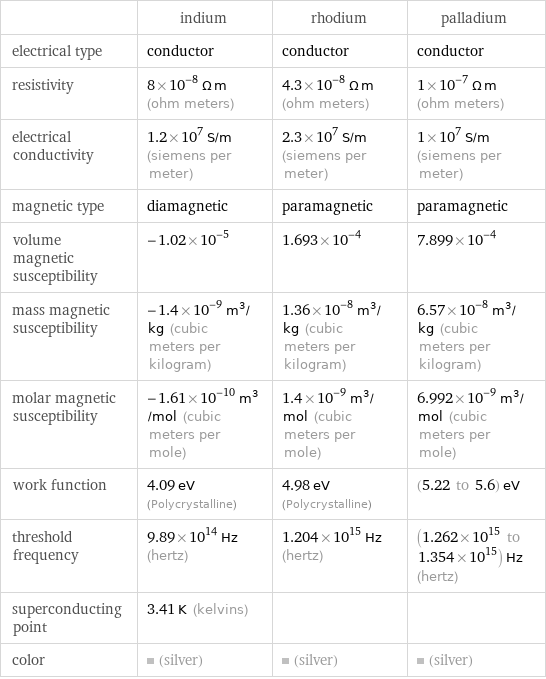
| indium | rhodium | palladium electrical type | conductor | conductor | conductor resistivity | 8×10^-8 Ω m (ohm meters) | 4.3×10^-8 Ω m (ohm meters) | 1×10^-7 Ω m (ohm meters) electrical conductivity | 1.2×10^7 S/m (siemens per meter) | 2.3×10^7 S/m (siemens per meter) | 1×10^7 S/m (siemens per meter) magnetic type | diamagnetic | paramagnetic | paramagnetic volume magnetic susceptibility | -1.02×10^-5 | 1.693×10^-4 | 7.899×10^-4 mass magnetic susceptibility | -1.4×10^-9 m^3/kg (cubic meters per kilogram) | 1.36×10^-8 m^3/kg (cubic meters per kilogram) | 6.57×10^-8 m^3/kg (cubic meters per kilogram) molar magnetic susceptibility | -1.61×10^-10 m^3/mol (cubic meters per mole) | 1.4×10^-9 m^3/mol (cubic meters per mole) | 6.992×10^-9 m^3/mol (cubic meters per mole) work function | 4.09 eV (Polycrystalline) | 4.98 eV (Polycrystalline) | (5.22 to 5.6) eV threshold frequency | 9.89×10^14 Hz (hertz) | 1.204×10^15 Hz (hertz) | (1.262×10^15 to 1.354×10^15) Hz (hertz) superconducting point | 3.41 K (kelvins) | | color | (silver) | (silver) | (silver)
Reactivity
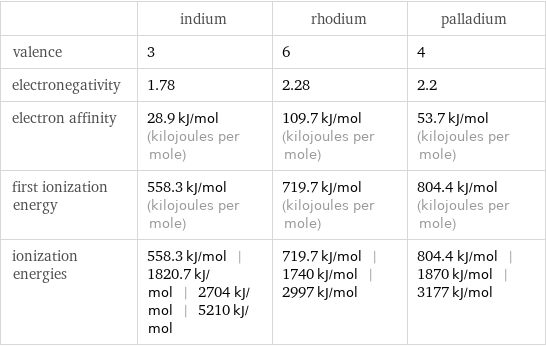
| indium | rhodium | palladium valence | 3 | 6 | 4 electronegativity | 1.78 | 2.28 | 2.2 electron affinity | 28.9 kJ/mol (kilojoules per mole) | 109.7 kJ/mol (kilojoules per mole) | 53.7 kJ/mol (kilojoules per mole) first ionization energy | 558.3 kJ/mol (kilojoules per mole) | 719.7 kJ/mol (kilojoules per mole) | 804.4 kJ/mol (kilojoules per mole) ionization energies | 558.3 kJ/mol | 1820.7 kJ/mol | 2704 kJ/mol | 5210 kJ/mol | 719.7 kJ/mol | 1740 kJ/mol | 2997 kJ/mol | 804.4 kJ/mol | 1870 kJ/mol | 3177 kJ/mol
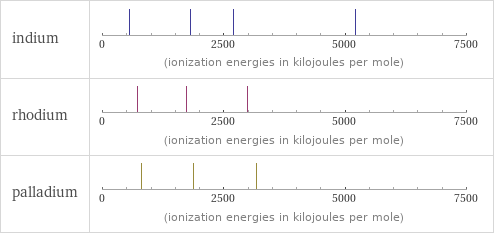
Reactivity
Atomic properties

| indium | rhodium | palladium term symbol | ^2P_(1/2) | ^4F_(9/2) | ^1S_0 atomic radius | 155 pm | 135 pm | 140 pm covalent radius | 142 pm | 142 pm | 139 pm van der Waals radius | 193 pm | | 163 pm (electronic ground state properties)
Abundances
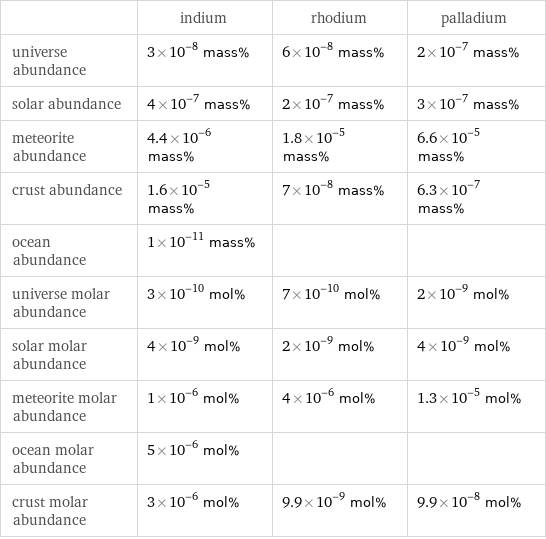
| indium | rhodium | palladium universe abundance | 3×10^-8 mass% | 6×10^-8 mass% | 2×10^-7 mass% solar abundance | 4×10^-7 mass% | 2×10^-7 mass% | 3×10^-7 mass% meteorite abundance | 4.4×10^-6 mass% | 1.8×10^-5 mass% | 6.6×10^-5 mass% crust abundance | 1.6×10^-5 mass% | 7×10^-8 mass% | 6.3×10^-7 mass% ocean abundance | 1×10^-11 mass% | | universe molar abundance | 3×10^-10 mol% | 7×10^-10 mol% | 2×10^-9 mol% solar molar abundance | 4×10^-9 mol% | 2×10^-9 mol% | 4×10^-9 mol% meteorite molar abundance | 1×10^-6 mol% | 4×10^-6 mol% | 1.3×10^-5 mol% ocean molar abundance | 5×10^-6 mol% | | crust molar abundance | 3×10^-6 mol% | 9.9×10^-9 mol% | 9.9×10^-8 mol%