Input interpretation

alkaline earth metals | molar volume
Summary

median | 29.59 cm^3/mol highest | 45 cm^3/mol (radium) lowest | 4.877 cm^3/mol (beryllium) distribution |
Units

Ranked values
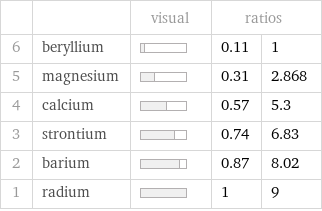
| | visual | ratios | 6 | beryllium | | 0.11 | 1 5 | magnesium | | 0.31 | 2.868 4 | calcium | | 0.57 | 5.3 3 | strontium | | 0.74 | 6.83 2 | barium | | 0.87 | 8.02 1 | radium | | 1 | 9
Molar volume rankings
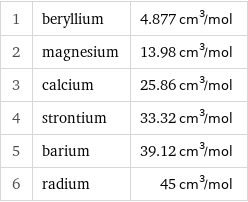
1 | beryllium | 4.877 cm^3/mol 2 | magnesium | 13.98 cm^3/mol 3 | calcium | 25.86 cm^3/mol 4 | strontium | 33.32 cm^3/mol 5 | barium | 39.12 cm^3/mol 6 | radium | 45 cm^3/mol
Unit conversions for median molar volume 29.59 cm^3/mol
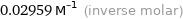
0.02959 M^(-1) (inverse molar)
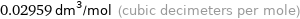
0.02959 dm^3/mol (cubic decimeters per mole)
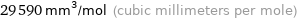
29590 mm^3/mol (cubic millimeters per mole)
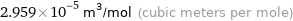
2.959×10^-5 m^3/mol (cubic meters per mole)
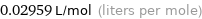
0.02959 L/mol (liters per mole)
Comparison for median molar volume 29.59 cm^3/mol
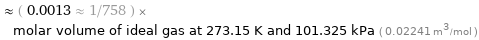
≈ ( 0.0013 ≈ 1/758 ) × molar volume of ideal gas at 273.15 K and 101.325 kPa ( 0.02241 m^3/mol )