Input interpretation
halogens | molar heat of vaporization
Summary
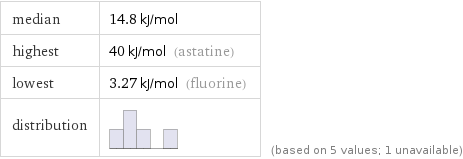
median | 14.8 kJ/mol highest | 40 kJ/mol (astatine) lowest | 3.27 kJ/mol (fluorine) distribution | | (based on 5 values; 1 unavailable)
Entity with missing value
tennessine
Ranked values
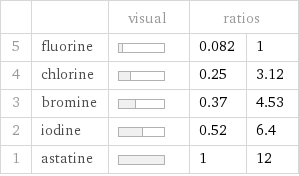
| | visual | ratios | 5 | fluorine | | 0.082 | 1 4 | chlorine | | 0.25 | 3.12 3 | bromine | | 0.37 | 4.53 2 | iodine | | 0.52 | 6.4 1 | astatine | | 1 | 12
Molar heat of vaporization rankings
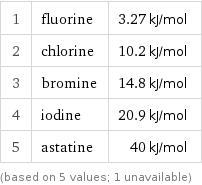
1 | fluorine | 3.27 kJ/mol 2 | chlorine | 10.2 kJ/mol 3 | bromine | 14.8 kJ/mol 4 | iodine | 20.9 kJ/mol 5 | astatine | 40 kJ/mol (based on 5 values; 1 unavailable)
Unit conversions for median molar heat of vaporization 14.8 kJ/mol
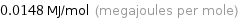
0.0148 MJ/mol (megajoules per mole)
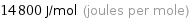
14800 J/mol (joules per mole)
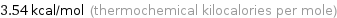
3.54 kcal/mol (thermochemical kilocalories per mole)

3540 cal/mol (calories per mole)

0.153 eV (molar electronvolts)