Input interpretation
lead(II) hydroxide | mercury(I) fluoride
Chemical names and formulas
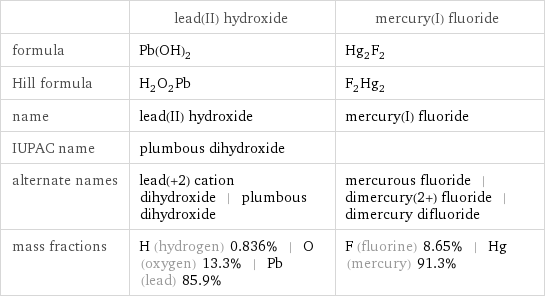
| lead(II) hydroxide | mercury(I) fluoride formula | Pb(OH)_2 | Hg_2F_2 Hill formula | H_2O_2Pb | F_2Hg_2 name | lead(II) hydroxide | mercury(I) fluoride IUPAC name | plumbous dihydroxide | alternate names | lead(+2) cation dihydroxide | plumbous dihydroxide | mercurous fluoride | dimercury(2+) fluoride | dimercury difluoride mass fractions | H (hydrogen) 0.836% | O (oxygen) 13.3% | Pb (lead) 85.9% | F (fluorine) 8.65% | Hg (mercury) 91.3%
Structure diagrams

Structure diagrams
Basic properties
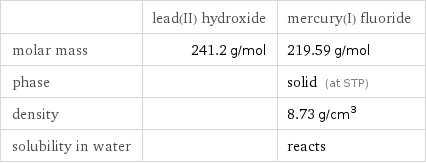
| lead(II) hydroxide | mercury(I) fluoride molar mass | 241.2 g/mol | 219.59 g/mol phase | | solid (at STP) density | | 8.73 g/cm^3 solubility in water | | reacts
Units
