Input interpretation
stannous sulfate | hexaamminecobalt(III) nitrate
Chemical names and formulas
_3 Hill formula | O_4SSn | CoH_18N_9O_9 name | stannous sulfate | hexaamminecobalt(III) nitrate IUPAC name | tin(+2) cation sulfate | cobaltic ammonia trinitrate alternate names | stannous sulphate | tin(2+) sulfate | tin(II) sulfate | tin(II) sulphate | azane; cobalt(+3) cation; trinitrate | cobaltic ammonia trinitrate mass fractions | O (oxygen) 29.8% | S (sulfur) 14.9% | Sn (tin) 55.3% | Co (cobalt) 17% | H (hydrogen) 5.23% | N (nitrogen) 36.3% | O (oxygen) 41.5%](../image_source/838f48201636f7e11cb1d578cbd91065.png)
| stannous sulfate | hexaamminecobalt(III) nitrate formula | SnSO_4 | [Co(NH_3)_6](NO_3)_3 Hill formula | O_4SSn | CoH_18N_9O_9 name | stannous sulfate | hexaamminecobalt(III) nitrate IUPAC name | tin(+2) cation sulfate | cobaltic ammonia trinitrate alternate names | stannous sulphate | tin(2+) sulfate | tin(II) sulfate | tin(II) sulphate | azane; cobalt(+3) cation; trinitrate | cobaltic ammonia trinitrate mass fractions | O (oxygen) 29.8% | S (sulfur) 14.9% | Sn (tin) 55.3% | Co (cobalt) 17% | H (hydrogen) 5.23% | N (nitrogen) 36.3% | O (oxygen) 41.5%
Structure diagrams
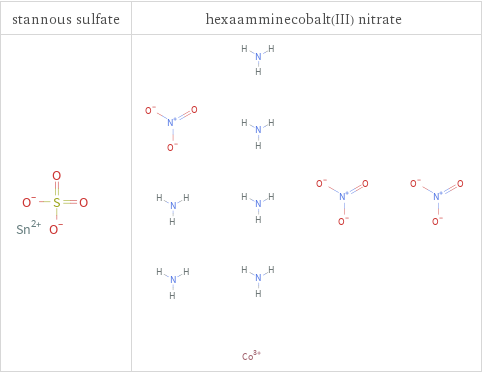
Structure diagrams
Basic properties

| stannous sulfate | hexaamminecobalt(III) nitrate molar mass | 214.77 g/mol | 347.13 g/mol phase | | solid (at STP) melting point | | 302.1 °C density | 4.15 g/cm^3 | solubility in water | soluble |
Units
