Input interpretation
type I cations
Members

hydrogen cation | lithium cation | beryllium cation | sodium cation | magnesium cation | aluminum cation | potassium cation | calcium cation | zinc cation | strontium cation | silver(I) cation | cadmium cation | cesium cation | barium cation (total: 14)
Ionic radii
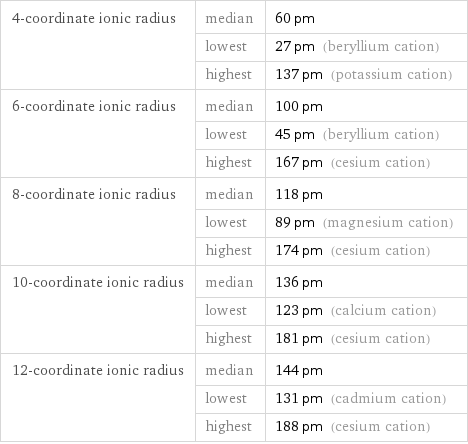
4-coordinate ionic radius | median | 60 pm | lowest | 27 pm (beryllium cation) | highest | 137 pm (potassium cation) 6-coordinate ionic radius | median | 100 pm | lowest | 45 pm (beryllium cation) | highest | 167 pm (cesium cation) 8-coordinate ionic radius | median | 118 pm | lowest | 89 pm (magnesium cation) | highest | 174 pm (cesium cation) 10-coordinate ionic radius | median | 136 pm | lowest | 123 pm (calcium cation) | highest | 181 pm (cesium cation) 12-coordinate ionic radius | median | 144 pm | lowest | 131 pm (cadmium cation) | highest | 188 pm (cesium cation)
Units
