Input interpretation

group 10 elements | atomic radius
Results
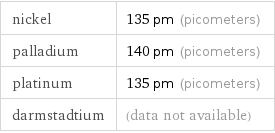
nickel | 135 pm (picometers) palladium | 140 pm (picometers) platinum | 135 pm (picometers) darmstadtium | (data not available)
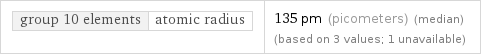
group 10 elements | atomic radius | 135 pm (picometers) (median) (based on 3 values; 1 unavailable)
Ranked values

| | visual | ratios | 3 | nickel | | 0.96 | 1 2 | platinum | | 0.96 | 1 1 | palladium | | 1 | 1.04
Plot
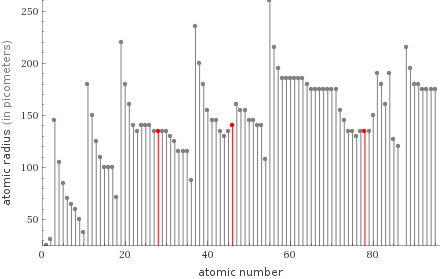
Plot
Periodic table location
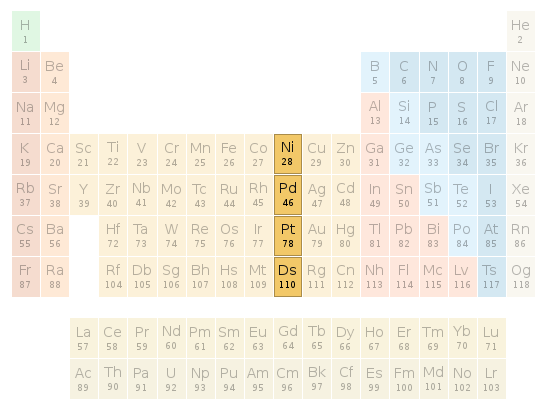
Periodic table location
Atomic radii properties
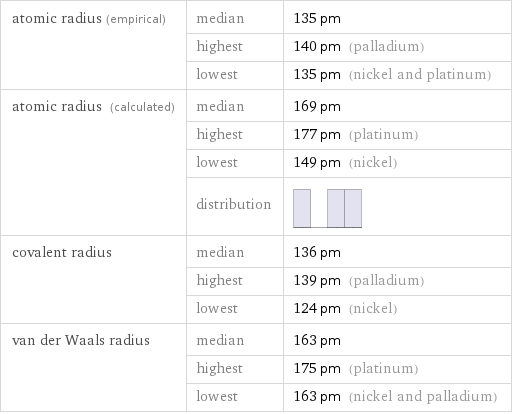
atomic radius (empirical) | median | 135 pm | highest | 140 pm (palladium) | lowest | 135 pm (nickel and platinum) atomic radius (calculated) | median | 169 pm | highest | 177 pm (platinum) | lowest | 149 pm (nickel) | distribution | covalent radius | median | 136 pm | highest | 139 pm (palladium) | lowest | 124 pm (nickel) van der Waals radius | median | 163 pm | highest | 175 pm (platinum) | lowest | 163 pm (nickel and palladium)
Units

Unit conversions for median atomic radius 135 pm
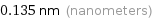
0.135 nm (nanometers)
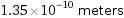
1.35×10^-10 meters
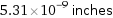
5.31×10^-9 inches
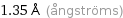
1.35 Å (ångströms)
Comparisons for median atomic radius 135 pm
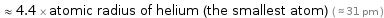
≈ 4.4 × atomic radius of helium (the smallest atom) ( ≈ 31 pm )
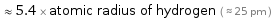
≈ 5.4 × atomic radius of hydrogen ( ≈ 25 pm )