Input interpretation
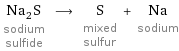
Na_2S sodium sulfide ⟶ S mixed sulfur + Na sodium
Balanced equation

Balance the chemical equation algebraically: Na_2S ⟶ S + Na Add stoichiometric coefficients, c_i, to the reactants and products: c_1 Na_2S ⟶ c_2 S + c_3 Na Set the number of atoms in the reactants equal to the number of atoms in the products for Na and S: Na: | 2 c_1 = c_3 S: | c_1 = c_2 Since the coefficients are relative quantities and underdetermined, choose a coefficient to set arbitrarily. To keep the coefficients small, the arbitrary value is ordinarily one. For instance, set c_1 = 1 and solve the system of equations for the remaining coefficients: c_1 = 1 c_2 = 1 c_3 = 2 Substitute the coefficients into the chemical reaction to obtain the balanced equation: Answer: | | Na_2S ⟶ S + 2 Na
Structures

⟶ +
Names
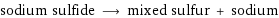
sodium sulfide ⟶ mixed sulfur + sodium
Equilibrium constant
![Construct the equilibrium constant, K, expression for: Na_2S ⟶ S + Na Plan: • Balance the chemical equation. • Determine the stoichiometric numbers. • Assemble the activity expression for each chemical species. • Use the activity expressions to build the equilibrium constant expression. Write the balanced chemical equation: Na_2S ⟶ S + 2 Na Assign stoichiometric numbers, ν_i, using the stoichiometric coefficients, c_i, from the balanced chemical equation in the following manner: ν_i = -c_i for reactants and ν_i = c_i for products: chemical species | c_i | ν_i Na_2S | 1 | -1 S | 1 | 1 Na | 2 | 2 Assemble the activity expressions accounting for the state of matter and ν_i: chemical species | c_i | ν_i | activity expression Na_2S | 1 | -1 | ([Na2S])^(-1) S | 1 | 1 | [S] Na | 2 | 2 | ([Na])^2 The equilibrium constant symbol in the concentration basis is: K_c Mulitply the activity expressions to arrive at the K_c expression: Answer: | | K_c = ([Na2S])^(-1) [S] ([Na])^2 = ([S] ([Na])^2)/([Na2S])](../image_source/474548376a5494e884897af7d92b189a.png)
Construct the equilibrium constant, K, expression for: Na_2S ⟶ S + Na Plan: • Balance the chemical equation. • Determine the stoichiometric numbers. • Assemble the activity expression for each chemical species. • Use the activity expressions to build the equilibrium constant expression. Write the balanced chemical equation: Na_2S ⟶ S + 2 Na Assign stoichiometric numbers, ν_i, using the stoichiometric coefficients, c_i, from the balanced chemical equation in the following manner: ν_i = -c_i for reactants and ν_i = c_i for products: chemical species | c_i | ν_i Na_2S | 1 | -1 S | 1 | 1 Na | 2 | 2 Assemble the activity expressions accounting for the state of matter and ν_i: chemical species | c_i | ν_i | activity expression Na_2S | 1 | -1 | ([Na2S])^(-1) S | 1 | 1 | [S] Na | 2 | 2 | ([Na])^2 The equilibrium constant symbol in the concentration basis is: K_c Mulitply the activity expressions to arrive at the K_c expression: Answer: | | K_c = ([Na2S])^(-1) [S] ([Na])^2 = ([S] ([Na])^2)/([Na2S])
Rate of reaction
![Construct the rate of reaction expression for: Na_2S ⟶ S + Na Plan: • Balance the chemical equation. • Determine the stoichiometric numbers. • Assemble the rate term for each chemical species. • Write the rate of reaction expression. Write the balanced chemical equation: Na_2S ⟶ S + 2 Na Assign stoichiometric numbers, ν_i, using the stoichiometric coefficients, c_i, from the balanced chemical equation in the following manner: ν_i = -c_i for reactants and ν_i = c_i for products: chemical species | c_i | ν_i Na_2S | 1 | -1 S | 1 | 1 Na | 2 | 2 The rate term for each chemical species, B_i, is 1/ν_i(Δ[B_i])/(Δt) where [B_i] is the amount concentration and t is time: chemical species | c_i | ν_i | rate term Na_2S | 1 | -1 | -(Δ[Na2S])/(Δt) S | 1 | 1 | (Δ[S])/(Δt) Na | 2 | 2 | 1/2 (Δ[Na])/(Δt) (for infinitesimal rate of change, replace Δ with d) Set the rate terms equal to each other to arrive at the rate expression: Answer: | | rate = -(Δ[Na2S])/(Δt) = (Δ[S])/(Δt) = 1/2 (Δ[Na])/(Δt) (assuming constant volume and no accumulation of intermediates or side products)](../image_source/1f8c1b77e97e9a59bae3350a9b724f9b.png)
Construct the rate of reaction expression for: Na_2S ⟶ S + Na Plan: • Balance the chemical equation. • Determine the stoichiometric numbers. • Assemble the rate term for each chemical species. • Write the rate of reaction expression. Write the balanced chemical equation: Na_2S ⟶ S + 2 Na Assign stoichiometric numbers, ν_i, using the stoichiometric coefficients, c_i, from the balanced chemical equation in the following manner: ν_i = -c_i for reactants and ν_i = c_i for products: chemical species | c_i | ν_i Na_2S | 1 | -1 S | 1 | 1 Na | 2 | 2 The rate term for each chemical species, B_i, is 1/ν_i(Δ[B_i])/(Δt) where [B_i] is the amount concentration and t is time: chemical species | c_i | ν_i | rate term Na_2S | 1 | -1 | -(Δ[Na2S])/(Δt) S | 1 | 1 | (Δ[S])/(Δt) Na | 2 | 2 | 1/2 (Δ[Na])/(Δt) (for infinitesimal rate of change, replace Δ with d) Set the rate terms equal to each other to arrive at the rate expression: Answer: | | rate = -(Δ[Na2S])/(Δt) = (Δ[S])/(Δt) = 1/2 (Δ[Na])/(Δt) (assuming constant volume and no accumulation of intermediates or side products)
Chemical names and formulas
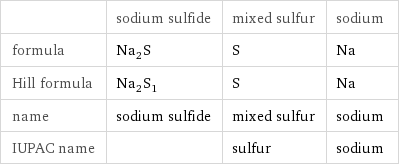
| sodium sulfide | mixed sulfur | sodium formula | Na_2S | S | Na Hill formula | Na_2S_1 | S | Na name | sodium sulfide | mixed sulfur | sodium IUPAC name | | sulfur | sodium
Substance properties
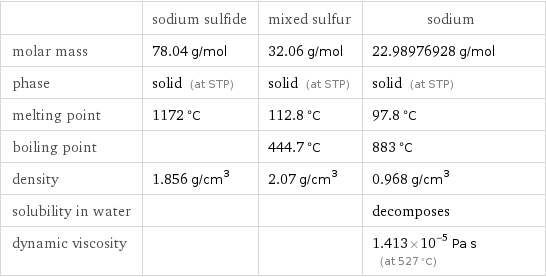
| sodium sulfide | mixed sulfur | sodium molar mass | 78.04 g/mol | 32.06 g/mol | 22.98976928 g/mol phase | solid (at STP) | solid (at STP) | solid (at STP) melting point | 1172 °C | 112.8 °C | 97.8 °C boiling point | | 444.7 °C | 883 °C density | 1.856 g/cm^3 | 2.07 g/cm^3 | 0.968 g/cm^3 solubility in water | | | decomposes dynamic viscosity | | | 1.413×10^-5 Pa s (at 527 °C)
Units
