Input interpretation
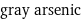
gray arsenic
Chemical names and formulas

formula | As name | gray arsenic IUPAC name | arsenic alternate names | arsenic gray | crystalline rhombohedral arsenic | metallic arsenic mass fractions | As (arsenic) 100%
Structure diagram

Structure diagram
Basic properties
molar mass | 74.921595 g/mol phase | solid (at STP) melting point | 817 °C boiling point | 616 °C density | 5.727 g/cm^3 solubility in water | insoluble
Units

Solid properties (at STP)
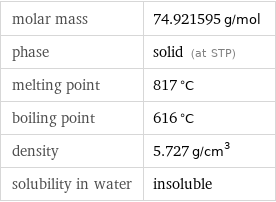
density | 5.727 g/cm^3 vapor pressure | 3.1 mmHg (at 400 °C) refractive index | 1.001552
Units

Thermodynamic properties
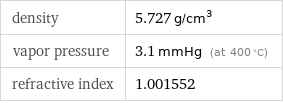
specific heat capacity c_p | gas | 0.2776 J/(g K) | solid | 0.3283 J/(g K) molar heat capacity c_p | gas | 20.8 J/(mol K) | solid | 24.6 J/(mol K) specific free energy of formation Δ_fG° | gas | 3.484 kJ/g molar free energy of formation Δ_fG° | gas | 261 kJ/mol specific heat of formation Δ_fH° | gas | 4.038 kJ/g | solid | 0 kJ/g molar heat of formation Δ_fH° | gas | 302.5 kJ/mol | solid | 0 kJ/mol specific entropy S° | gas | 2.325 J/(g K) | solid | 0.4685 J/(g K) molar entropy S° | gas | 174.2 J/(mol K) | solid | 35.1 J/(mol K) molar heat of fusion | 24.44 kJ/mol | specific heat of fusion | 0.3262 kJ/g | critical temperature | 1673 K | (at STP)
Chemical identifiers
![CAS number | 7440-38-2 PubChem CID number | 5359596 PubChem SID number | 24852109 SMILES identifier | [As] InChI identifier | InChI=1/As RTECS number | CG0525000 MDL number | MFCD00085309](../image_source/70969cb29ae4e1cbc42714fb2dcd0d64.png)
CAS number | 7440-38-2 PubChem CID number | 5359596 PubChem SID number | 24852109 SMILES identifier | [As] InChI identifier | InChI=1/As RTECS number | CG0525000 MDL number | MFCD00085309
NFPA label

NFPA label

NFPA health rating | 3 NFPA fire rating | 2
Toxicity properties

odor | odorless

RTECS classes | tumorigen | drug | mutagen | reproductive effector | human data