Input interpretation
lithium hydroxide | sodium chloride
Chemical names and formulas
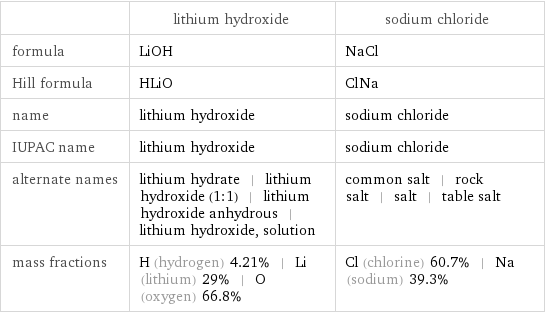
| lithium hydroxide | sodium chloride formula | LiOH | NaCl Hill formula | HLiO | ClNa name | lithium hydroxide | sodium chloride IUPAC name | lithium hydroxide | sodium chloride alternate names | lithium hydrate | lithium hydroxide (1:1) | lithium hydroxide anhydrous | lithium hydroxide, solution | common salt | rock salt | salt | table salt mass fractions | H (hydrogen) 4.21% | Li (lithium) 29% | O (oxygen) 66.8% | Cl (chlorine) 60.7% | Na (sodium) 39.3%
Structure diagrams
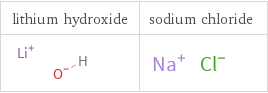
Structure diagrams
Basic properties
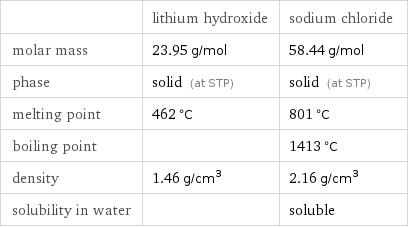
| lithium hydroxide | sodium chloride molar mass | 23.95 g/mol | 58.44 g/mol phase | solid (at STP) | solid (at STP) melting point | 462 °C | 801 °C boiling point | | 1413 °C density | 1.46 g/cm^3 | 2.16 g/cm^3 solubility in water | | soluble
Units
