Input interpretation
sulfuric acid
Physical properties
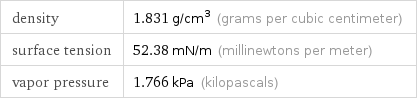
density | 1.831 g/cm^3 (grams per cubic centimeter) surface tension | 52.38 mN/m (millinewtons per meter) vapor pressure | 1.766 kPa (kilopascals)
Solution properties
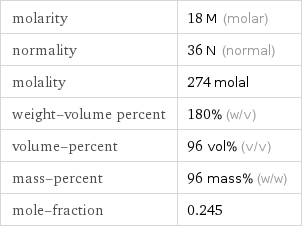
molarity | 18 M (molar) normality | 36 N (normal) molality | 274 molal weight-volume percent | 180% (w/v) volume-percent | 96 vol% (v/v) mass-percent | 96 mass% (w/w) mole-fraction | 0.245
Solute properties per 100 mL

| sulfuric acid molar amount | 1.8 mol (moles) mass | 0.177 kg (kilograms) equivalents | 3.6 eq (equivalents) of hydrogen cation 1.8 eq (equivalents) of sulfate anion
Acid-base information
![K_a | 4.17×10^6 pK_a | -6.62 pH | -1.26 [H_3O^+] | 18 mol/L (moles per liter) pOH | 15.3 [OH^-] | 5.56×10^-16 mol/L (moles per liter) % ionization | 100%](../image_source/258efa414578cd0c5f5bb644b88ca05f.png)
K_a | 4.17×10^6 pK_a | -6.62 pH | -1.26 [H_3O^+] | 18 mol/L (moles per liter) pOH | 15.3 [OH^-] | 5.56×10^-16 mol/L (moles per liter) % ionization | 100%
Lewis structure

Draw the Lewis structure of sulfuric acid. Start by drawing the overall structure of the molecule, ignoring potential double and triple bonds: Count the total valence electrons of the hydrogen (n_H, val = 1), oxygen (n_O, val = 6), and sulfur (n_S, val = 6) atoms: 2 n_H, val + 4 n_O, val + n_S, val = 32 Calculate the number of electrons needed to completely fill the valence shells for hydrogen (n_H, full = 2), oxygen (n_O, full = 8), and sulfur (n_S, full = 8): 2 n_H, full + 4 n_O, full + n_S, full = 44 Subtracting these two numbers shows that 44 - 32 = 12 bonding electrons are needed. Each bond has two electrons, so we expect that the above diagram has all the necessary bonds. However, to minimize formal charge oxygen wants 2 bonds. Identify the atoms that want additional bonds and the number of electrons remaining on each atom: In order to minimize their formal charge, atoms with large electronegativities can force atoms with smaller electronegativities on period 3 or higher to expand their valence shells. The electronegativities of the atoms are 2.20 (hydrogen), 2.58 (sulfur), and 3.44 (oxygen). Because the electronegativity of sulfur is smaller than the electronegativity of oxygen, expand the valence shell of sulfur to 6 bonds. Therefore we add a total of 2 bonds to the diagram: Answer: | |
Chemical names and formulas

formula | H_2SO_4 Hill formula | H_2O_4S name | sulfuric acid IUPAC name | sulfuric acid