Input interpretation

aqueous compounds | freezing point
Summary

median | -24 °C highest | 3 °C (aqueous sulfuric acid) lowest | -57.5 °C (ammonium hydroxide) distribution | | (based on 8 values; 2 unavailable)
Entities with missing values
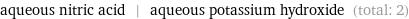
aqueous nitric acid | aqueous potassium hydroxide (total: 2)
Freezing point rankings
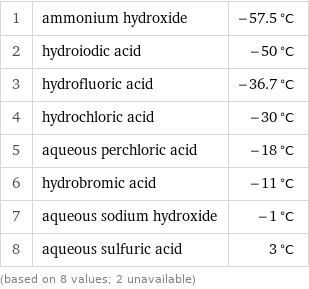
1 | ammonium hydroxide | -57.5 °C 2 | hydroiodic acid | -50 °C 3 | hydrofluoric acid | -36.7 °C 4 | hydrochloric acid | -30 °C 5 | aqueous perchloric acid | -18 °C 6 | hydrobromic acid | -11 °C 7 | aqueous sodium hydroxide | -1 °C 8 | aqueous sulfuric acid | 3 °C (based on 8 values; 2 unavailable)
Unit conversions for median freezing point -24 °C
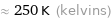
≈ 250 K (kelvins)

≈ -11 °F (degrees Fahrenheit)
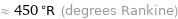
≈ 450 °R (degrees Rankine)
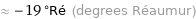
≈ -19 °Ré (degrees Réaumur)
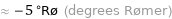
≈ -5 °Rø (degrees Rømer)
Comparison for median freezing point -24 °C

40 °C below temperature at STP (standard temperature and pressure), using the convention of European and South American natural gas companies (15 °C)
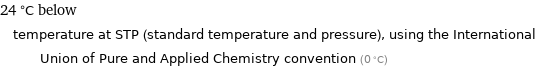
24 °C below temperature at STP (standard temperature and pressure), using the International Union of Pure and Applied Chemistry convention (0 °C)
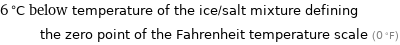
6 °C below temperature of the ice/salt mixture defining the zero point of the Fahrenheit temperature scale (0 °F)
Corresponding quantities

Thermodynamic energy E from E = kT: | 21 meV (millielectronvolts)
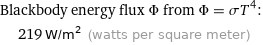
Blackbody energy flux Φ from Φ = σT^4: | 219 W/m^2 (watts per square meter)

Approximate luminous exitance from a planar blackbody radiator perpendicular to its surface: | 3.3×10^-27 lx (lux)