Input interpretation
scandium chloride
Chemical names and formulas

formula | ScCl_3 Hill formula | Cl_3Sc name | scandium chloride IUPAC name | trichloroscandium alternate names | scandium(3+) chloride | scandium trichloride | trichloroscandium mass fractions | Cl (chlorine) 70.3% | Sc (scandium) 29.7%
Structure diagram
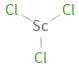
Structure diagram

| bond counts | bond lengths | 3 bonds | 2.2 Å
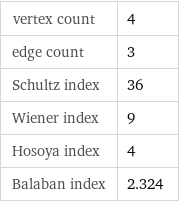
vertex count | 4 edge count | 3 Schultz index | 36 Wiener index | 9 Hosoya index | 4 Balaban index | 2.324
3D structure

3D structure
Basic properties
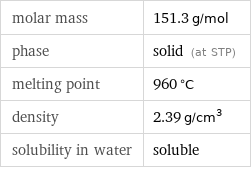
molar mass | 151.3 g/mol phase | solid (at STP) melting point | 960 °C density | 2.39 g/cm^3 solubility in water | soluble
Units

Solid properties (at STP)

density | 2.39 g/cm^3
Units

Thermodynamic properties
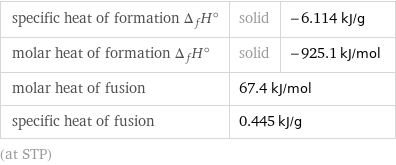
specific heat of formation Δ_fH° | solid | -6.114 kJ/g molar heat of formation Δ_fH° | solid | -925.1 kJ/mol molar heat of fusion | 67.4 kJ/mol | specific heat of fusion | 0.445 kJ/g | (at STP)
Chemical identifiers
Cl InChI identifier | InChI=1/3ClH.Sc/h3*1H;/q;;;+3/p-3/f3Cl.Sc/h3*1h;/q3*-1;m RTECS number | VQ8925000 MDL number | MFCD00011221](../image_source/dc56928bb1420058668761cb71e1a44e.png)
CAS number | 10361-84-9 PubChem CID number | 82586 PubChem SID number | 24865640 SMILES identifier | Cl[Sc](Cl)Cl InChI identifier | InChI=1/3ClH.Sc/h3*1H;/q;;;+3/p-3/f3Cl.Sc/h3*1h;/q3*-1;m RTECS number | VQ8925000 MDL number | MFCD00011221
Toxicity properties

RTECS classes | other