Input interpretation
actinium (chemical element)
Periodic table location
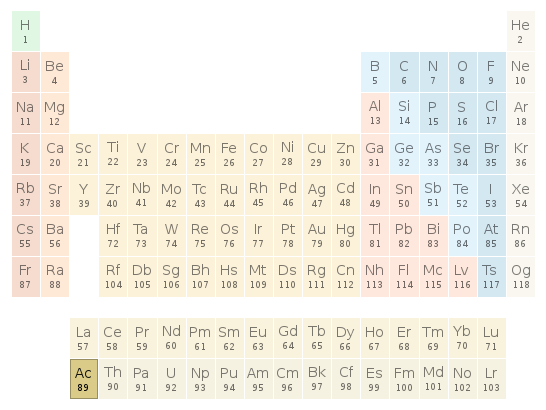
Periodic table location
Image

Image
Basic elemental properties
![atomic symbol | Ac atomic number | 89 short electronic configuration | [Rn]7s^26d^1 Aufbau diagram | 6d 7s block | f period | 7 atomic mass | 227 u (unified atomic mass units) (atomic mass number given for longest-lived isotope) half-life | 21.787 years](../image_source/6924e3baa62268d497df8d7e31670518.png)
atomic symbol | Ac atomic number | 89 short electronic configuration | [Rn]7s^26d^1 Aufbau diagram | 6d 7s block | f period | 7 atomic mass | 227 u (unified atomic mass units) (atomic mass number given for longest-lived isotope) half-life | 21.787 years
Thermodynamic properties
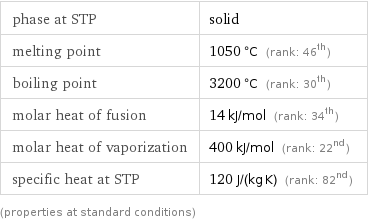
phase at STP | solid melting point | 1050 °C (rank: 46th) boiling point | 3200 °C (rank: 30th) molar heat of fusion | 14 kJ/mol (rank: 34th) molar heat of vaporization | 400 kJ/mol (rank: 22nd) specific heat at STP | 120 J/(kg K) (rank: 82nd) (properties at standard conditions)
Material properties
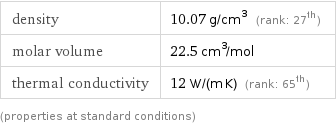
density | 10.07 g/cm^3 (rank: 27th) molar volume | 22.5 cm^3/mol thermal conductivity | 12 W/(m K) (rank: 65th) (properties at standard conditions)
Electromagnetic properties

color | (silver)
Reactivity
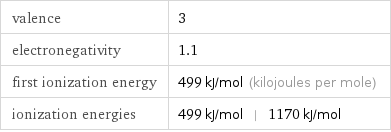
valence | 3 electronegativity | 1.1 first ionization energy | 499 kJ/mol (kilojoules per mole) ionization energies | 499 kJ/mol | 1170 kJ/mol
Reactivity
Atomic properties

term symbol | ^2D_(3/2) atomic radius | 195 pm covalent radius | 215 pm (electronic ground state properties)
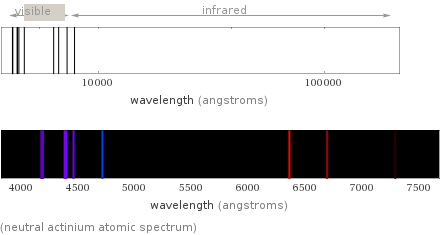
(neutral actinium atomic spectrum)
Abundances

solar abundance | 0 mass% human abundance | 0 mass% solar molar abundance | 0 mol% human molar abundance | 0 mol%
Nuclear properties

half-life | 21.787 yr lifetime | 31.431 yr (rank: 1st) specific radioactivity | 2.676 TBq/g decay mode | beta decay nuclear spin | Ac-206: 3^+ | Ac-207: 9/2^- | Ac-208: 3^+ | Ac-209: 9/2^- | Ac-210: 7^+ | Ac-211: 9/2^- | Ac-212: 6^+ | Ac-213: 9/2^- | Ac-214: 5^+ | Ac-215: 9/2^- | Ac-216: 1^- | Ac-217: 9/2^- | Ac-218: 1^- | Ac-219: 9/2^- | Ac-220: 3^- | Ac-221: 3/2^- | Ac-222: 1^- | Ac-223: 5/2^- | Ac-224: 0^- | Ac-225: 3/2^- | Ac-226: 1^- | Ac-227: 3/2^- | Ac-228: 3^+ | Ac-229: 3/2^+ | Ac-230: 1^+ | Ac-231: 1/2^+ | Ac-232: 1^+ | Ac-233: 1/2^+ | Ac-235: 1/2^+ unstable isotopes | Ac-227 (21.772 yr) | Ac-225 (10 days) | Ac-226 (29.36 h) | Ac-228 (6.14 h) | Ac-224 (170 min) | Ac-229 (62.67 min) | Ac-231 (7.5 min) | Ac-233 (145 s) | Ac-223 (126 s) | Ac-230 (122 s) | Ac-232 (119 s) | Ac-236 (100 s) | Ac-235 (60 s) | Ac-234 (44 s) | Ac-214 (8.2 s) | Ac-222 (5 s) | Ac-212 (930 ms) | Ac-213 (738 ms) | Ac-210 (350 ms) | Ac-211 (210 ms) | Ac-215 (170 ms) | Ac-209 (100 ms) | Ac-208 (95 ms) | Ac-221 (52 ms) | Ac-207 (27 ms) | Ac-220 (26.36 ms) | Ac-206 (22 ms) | Ac-216 (440 µs) | Ac-219 (11.8 µs) | Ac-218 (1.08 µs) | Ac-217 (69 ns) neutron cross-section | 880 b neutron mass absorption | 0.079 m^2/kg
Identifiers

CAS number | 7440-34-8 PubChem CID number | 23965