Input interpretation
cesium perchlorate
Chemical names and formulas

formula | CsClO_4 Hill formula | ClCsO_4 name | cesium perchlorate alternate names | caesium perchlorate | cesium perchlorate(metals basis) | perchloric acid, cesium salt mass fractions | Cl (chlorine) 15.3% | Cs (cesium) 57.2% | O (oxygen) 27.5%
Structure diagram
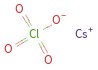
Structure diagram
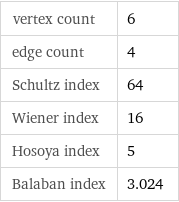
vertex count | 6 edge count | 4 Schultz index | 64 Wiener index | 16 Hosoya index | 5 Balaban index | 3.024
Basic properties
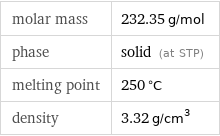
molar mass | 232.35 g/mol phase | solid (at STP) melting point | 250 °C density | 3.32 g/cm^3
Units

Solid properties (at STP)
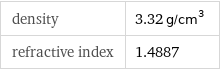
density | 3.32 g/cm^3 refractive index | 1.4887
Units

Thermodynamic properties
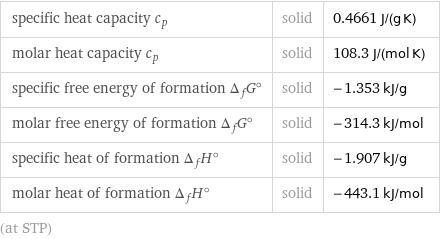
specific heat capacity c_p | solid | 0.4661 J/(g K) molar heat capacity c_p | solid | 108.3 J/(mol K) specific free energy of formation Δ_fG° | solid | -1.353 kJ/g molar free energy of formation Δ_fG° | solid | -314.3 kJ/mol specific heat of formation Δ_fH° | solid | -1.907 kJ/g molar heat of formation Δ_fH° | solid | -443.1 kJ/mol (at STP)
Chemical identifiers
![CAS number | 13454-84-7 PubChem CID number | 123309 PubChem SID number | 24858724 SMILES identifier | [O-]Cl(=O)(=O)=O.[Cs+] InChI identifier | InChI=1/ClHO4.Cs/c2-1(3, 4)5;/h(H, 2, 3, 4, 5);/q;+1/p-1/fClO4.Cs/q-1;m MDL number | MFCD00010956](../image_source/07d00bf49869f7bc0f81e709a2c905f6.png)
CAS number | 13454-84-7 PubChem CID number | 123309 PubChem SID number | 24858724 SMILES identifier | [O-]Cl(=O)(=O)=O.[Cs+] InChI identifier | InChI=1/ClHO4.Cs/c2-1(3, 4)5;/h(H, 2, 3, 4, 5);/q;+1/p-1/fClO4.Cs/q-1;m MDL number | MFCD00010956
NFPA label

NFPA label

NFPA hazards | oxidizing agent
Ion equivalents

Cs^+ (cesium cation) | 1 (ClO_4)^- (perchlorate anion) | 1