Input interpretation
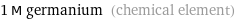
1 M germanium (chemical element)
Physical properties
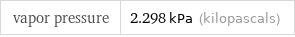
vapor pressure | 2.298 kPa (kilopascals)
Solution properties
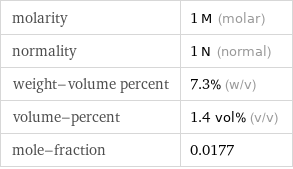
molarity | 1 M (molar) normality | 1 N (normal) weight-volume percent | 7.3% (w/v) volume-percent | 1.4 vol% (v/v) mole-fraction | 0.0177
Solute properties per 100 mL
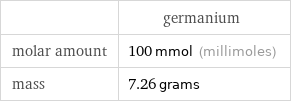
| germanium molar amount | 100 mmol (millimoles) mass | 7.26 grams
Preparation
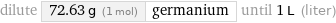
dilute 72.63 g (1 mol) | germanium until 1 L (liter)
Structure diagram
Structure diagram
Chemical names and formulas
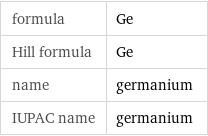
formula | Ge Hill formula | Ge name | germanium IUPAC name | germanium
Substance properties
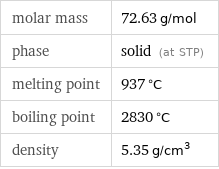
molar mass | 72.63 g/mol phase | solid (at STP) melting point | 937 °C boiling point | 2830 °C density | 5.35 g/cm^3
Units
