Input interpretation
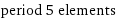
period 5 elements
Members

rubidium | strontium | yttrium | zirconium | niobium | molybdenum | technetium | ruthenium | rhodium | palladium | silver | cadmium | indium | tin | antimony | iodine | tellurium | xenon (total: 18)
Periodic table location
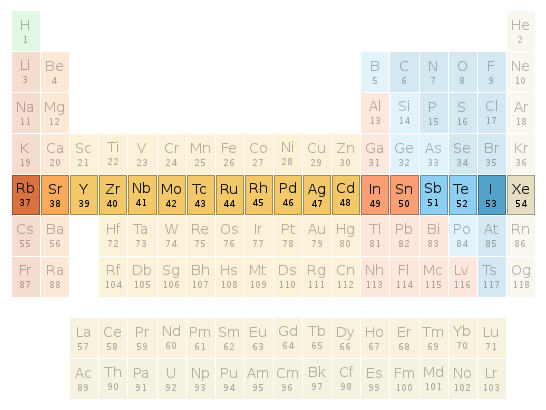
Periodic table location
Images

Images
Basic elemental properties

atomic symbol | all | Ag | Cd | I | In | Mo | Nb | Pd | Rb | Rh | Ru | Sb | Sn | Sr | Tc | Te | Xe | Y | Zr atomic number | median | 45.5 | highest | 54 (xenon) | lowest | 37 (rubidium) | distribution | block | all | d | p | s group | all | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 period | all | 5 atomic mass | median | 104.66 u | highest | 131.293 u (xenon) | lowest | 85.4678 u (rubidium) | distribution |
Thermodynamic properties

phase at STP | all | gas | solid melting point | median | 869 °C | highest | 2623 °C (molybdenum) | lowest | -111.8 °C (xenon) | distribution | boiling point | median | 2382 °C | highest | 4744 °C (niobium) | lowest | -108 °C (xenon) | distribution | critical temperature | median | 819 K | highest | 2093 K (rubidium) | lowest | 289.77 K (xenon) | distribution | critical pressure | median | 11.7 MPa | highest | 16 MPa (rubidium) | lowest | 5.841 MPa (xenon) | distribution | molar heat of fusion | median | 14 kJ/mol | highest | 36 kJ/mol (molybdenum) | lowest | 2.19 kJ/mol (rubidium) | distribution | molar heat of vaporization | median | 272 kJ/mol | highest | 690 kJ/mol (niobium) | lowest | 12.64 kJ/mol (xenon) | distribution | specific heat at STP | median | 239 J/(kg K) | highest | 429 J/(kg K) (iodine) | lowest | 63 J/(kg K) (technetium) | distribution | (properties at standard conditions)
Material properties

density | median | 7.31 g/cm^3 | highest | 12.45 g/cm^3 (rhodium) | lowest | 0.0059 g/cm^3 (xenon) | distribution | liquid density | median | 7.005 g/cm^3 | highest | 10.7 g/cm^3 (rhodium) | lowest | 1.46 g/cm^3 (rubidium) | distribution | molar volume | median | 14.86 cm^3/mol | highest | 22000 cm^3/mol (xenon) | lowest | 8.171 cm^3/mol (ruthenium) Mohs hardness | median | 2.7 | highest | 6.5 (between microcline and quartz) (ruthenium) | lowest | 0.3 (between talc and gypsum) (rubidium) | distribution | Vickers hardness | all | 251 MPa | 461 MPa | 903 MPa | 1246 MPa | 1320 MPa | 1530 MPa | 2300 MPa Brinell hardness | all | 0.216 MPa | 8.83 MPa | 24.5 MPa | 37.3 MPa | 51 MPa | 180 MPa | 203 MPa | 294 MPa | 589 MPa | 650 MPa | 736 MPa | 1100 MPa | 1500 MPa | 2160 MPa bulk modulus | median | 65 GPa | highest | 380 GPa (rhodium) | lowest | 2.5 GPa (rubidium) | distribution | shear modulus | median | 26 GPa | highest | 173 GPa (ruthenium) | lowest | 6.1 GPa (strontium) | distribution | Young's modulus | median | 66 GPa | highest | 447 GPa (ruthenium) | lowest | 2.4 GPa (rubidium) | distribution | Poisson ratio | all | 0.24 | 0.26 | 0.28 | 0.3 | 0.31 | 0.34 | 0.36 | 0.37 | 0.39 | 0.4 tensile yield strength | median | 230 MPa | highest | 1.29 GPa (technetium) | lowest | 67 MPa (yttrium) ultimate tensile strength | all | 4 MPa | 11 MPa | 75 MPa | 140 MPa | 150 MPa | 180 MPa | 220 MPa | 330 MPa | 1.51 GPa sound speed | median | 3070 m/s | highest | 6190 m/s (molybdenum) | lowest | 1090 m/s (xenon) | distribution | thermal expansion | median | 1.1×10^-5 K^(-1) | highest | 3.21×10^-5 K^(-1) (indium) | lowest | 4.8×10^-6 K^(-1) (molybdenum) | distribution | thermal conductivity | median | 56 W/(m K) | highest | 430 W/(m K) (silver) | lowest | 0.00565 W/(m K) (xenon) | distribution | (properties at standard conditions)