Input interpretation
molybdenum (chemical element)
Periodic table location
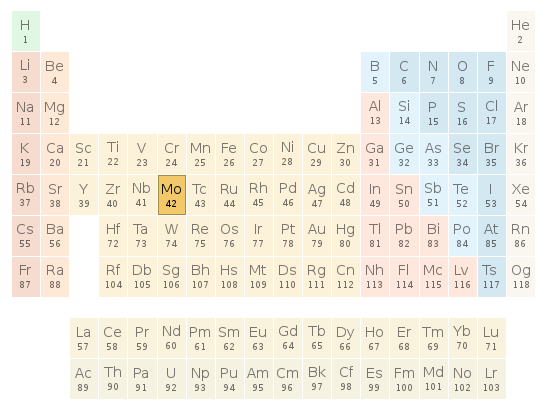
Periodic table location
Image

Image
Basic elemental properties
![atomic symbol | Mo atomic number | 42 short electronic configuration | [Kr]5s^14d^5 Aufbau diagram | 4d 5s block | d group | 6 period | 5 atomic mass | 95.95 u (unified atomic mass units)](../image_source/e1af24a8509418544ccaea5821865e4c.png)
atomic symbol | Mo atomic number | 42 short electronic configuration | [Kr]5s^14d^5 Aufbau diagram | 4d 5s block | d group | 6 period | 5 atomic mass | 95.95 u (unified atomic mass units)
Thermodynamic properties
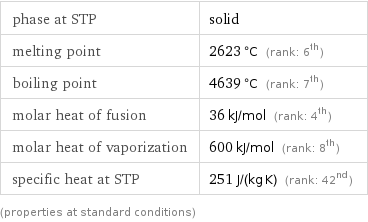
phase at STP | solid melting point | 2623 °C (rank: 6th) boiling point | 4639 °C (rank: 7th) molar heat of fusion | 36 kJ/mol (rank: 4th) molar heat of vaporization | 600 kJ/mol (rank: 8th) specific heat at STP | 251 J/(kg K) (rank: 42nd) (properties at standard conditions)
Material properties
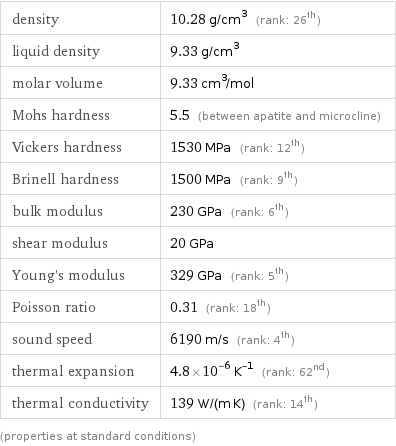
density | 10.28 g/cm^3 (rank: 26th) liquid density | 9.33 g/cm^3 molar volume | 9.33 cm^3/mol Mohs hardness | 5.5 (between apatite and microcline) Vickers hardness | 1530 MPa (rank: 12th) Brinell hardness | 1500 MPa (rank: 9th) bulk modulus | 230 GPa (rank: 6th) shear modulus | 20 GPa Young's modulus | 329 GPa (rank: 5th) Poisson ratio | 0.31 (rank: 18th) sound speed | 6190 m/s (rank: 4th) thermal expansion | 4.8×10^-6 K^(-1) (rank: 62nd) thermal conductivity | 139 W/(m K) (rank: 14th) (properties at standard conditions)
Electromagnetic properties
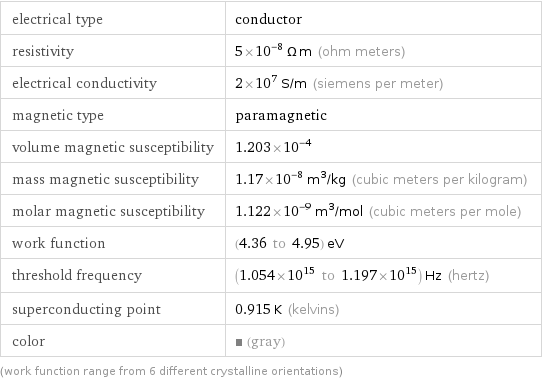
electrical type | conductor resistivity | 5×10^-8 Ω m (ohm meters) electrical conductivity | 2×10^7 S/m (siemens per meter) magnetic type | paramagnetic volume magnetic susceptibility | 1.203×10^-4 mass magnetic susceptibility | 1.17×10^-8 m^3/kg (cubic meters per kilogram) molar magnetic susceptibility | 1.122×10^-9 m^3/mol (cubic meters per mole) work function | (4.36 to 4.95) eV threshold frequency | (1.054×10^15 to 1.197×10^15) Hz (hertz) superconducting point | 0.915 K (kelvins) color | (gray) (work function range from 6 different crystalline orientations)
Reactivity
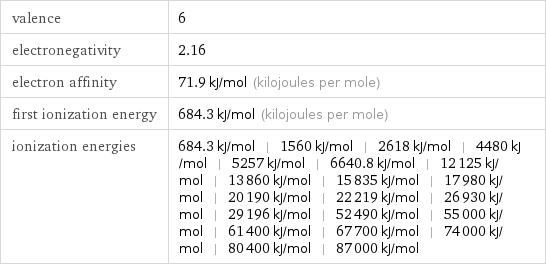
valence | 6 electronegativity | 2.16 electron affinity | 71.9 kJ/mol (kilojoules per mole) first ionization energy | 684.3 kJ/mol (kilojoules per mole) ionization energies | 684.3 kJ/mol | 1560 kJ/mol | 2618 kJ/mol | 4480 kJ/mol | 5257 kJ/mol | 6640.8 kJ/mol | 12125 kJ/mol | 13860 kJ/mol | 15835 kJ/mol | 17980 kJ/mol | 20190 kJ/mol | 22219 kJ/mol | 26930 kJ/mol | 29196 kJ/mol | 52490 kJ/mol | 55000 kJ/mol | 61400 kJ/mol | 67700 kJ/mol | 74000 kJ/mol | 80400 kJ/mol | 87000 kJ/mol
Reactivity
Atomic properties

term symbol | ^7S_3 atomic radius | 145 pm covalent radius | 154 pm (electronic ground state properties)
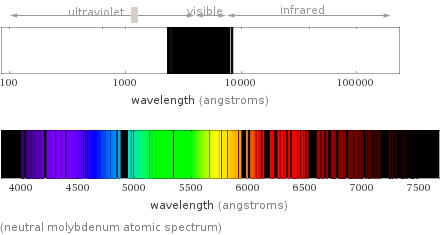
(neutral molybdenum atomic spectrum)
Abundances
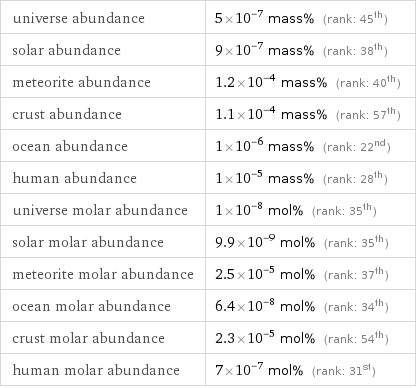
universe abundance | 5×10^-7 mass% (rank: 45th) solar abundance | 9×10^-7 mass% (rank: 38th) meteorite abundance | 1.2×10^-4 mass% (rank: 40th) crust abundance | 1.1×10^-4 mass% (rank: 57th) ocean abundance | 1×10^-6 mass% (rank: 22nd) human abundance | 1×10^-5 mass% (rank: 28th) universe molar abundance | 1×10^-8 mol% (rank: 35th) solar molar abundance | 9.9×10^-9 mol% (rank: 35th) meteorite molar abundance | 2.5×10^-5 mol% (rank: 37th) ocean molar abundance | 6.4×10^-8 mol% (rank: 34th) crust molar abundance | 2.3×10^-5 mol% (rank: 54th) human molar abundance | 7×10^-7 mol% (rank: 31st)
Nuclear properties
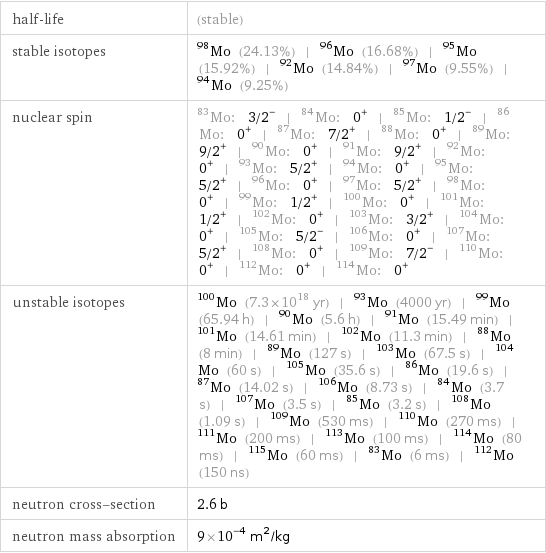
half-life | (stable) stable isotopes | Mo-98 (24.13%) | Mo-96 (16.68%) | Mo-95 (15.92%) | Mo-92 (14.84%) | Mo-97 (9.55%) | Mo-94 (9.25%) nuclear spin | Mo-83: 3/2^- | Mo-84: 0^+ | Mo-85: 1/2^- | Mo-86: 0^+ | Mo-87: 7/2^+ | Mo-88: 0^+ | Mo-89: 9/2^+ | Mo-90: 0^+ | Mo-91: 9/2^+ | Mo-92: 0^+ | Mo-93: 5/2^+ | Mo-94: 0^+ | Mo-95: 5/2^+ | Mo-96: 0^+ | Mo-97: 5/2^+ | Mo-98: 0^+ | Mo-99: 1/2^+ | Mo-100: 0^+ | Mo-101: 1/2^+ | Mo-102: 0^+ | Mo-103: 3/2^+ | Mo-104: 0^+ | Mo-105: 5/2^- | Mo-106: 0^+ | Mo-107: 5/2^+ | Mo-108: 0^+ | Mo-109: 7/2^- | Mo-110: 0^+ | Mo-112: 0^+ | Mo-114: 0^+ unstable isotopes | Mo-100 (7.3×10^18 yr) | Mo-93 (4000 yr) | Mo-99 (65.94 h) | Mo-90 (5.6 h) | Mo-91 (15.49 min) | Mo-101 (14.61 min) | Mo-102 (11.3 min) | Mo-88 (8 min) | Mo-89 (127 s) | Mo-103 (67.5 s) | Mo-104 (60 s) | Mo-105 (35.6 s) | Mo-86 (19.6 s) | Mo-87 (14.02 s) | Mo-106 (8.73 s) | Mo-84 (3.7 s) | Mo-107 (3.5 s) | Mo-85 (3.2 s) | Mo-108 (1.09 s) | Mo-109 (530 ms) | Mo-110 (270 ms) | Mo-111 (200 ms) | Mo-113 (100 ms) | Mo-114 (80 ms) | Mo-115 (60 ms) | Mo-83 (6 ms) | Mo-112 (150 ns) neutron cross-section | 2.6 b neutron mass absorption | 9×10^-4 m^2/kg
Identifiers

CAS number | 7439-98-7 PubChem CID number | 23932