Input interpretation
potassium chloride | rubidium nitrate
Chemical names and formulas
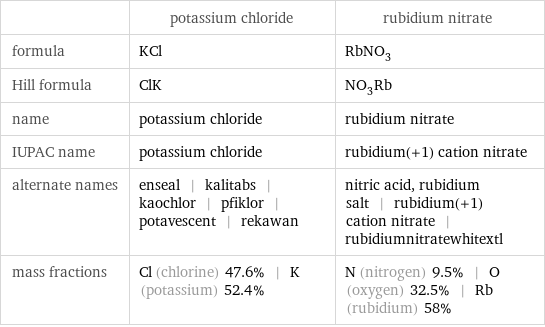
| potassium chloride | rubidium nitrate formula | KCl | RbNO_3 Hill formula | ClK | NO_3Rb name | potassium chloride | rubidium nitrate IUPAC name | potassium chloride | rubidium(+1) cation nitrate alternate names | enseal | kalitabs | kaochlor | pfiklor | potavescent | rekawan | nitric acid, rubidium salt | rubidium(+1) cation nitrate | rubidiumnitratewhitextl mass fractions | Cl (chlorine) 47.6% | K (potassium) 52.4% | N (nitrogen) 9.5% | O (oxygen) 32.5% | Rb (rubidium) 58%
Structure diagrams
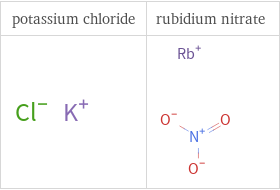
Structure diagrams
Basic properties
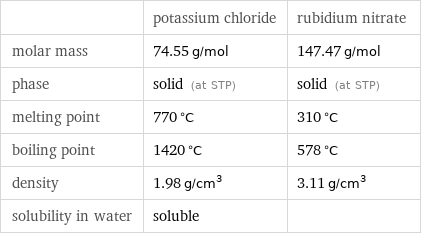
| potassium chloride | rubidium nitrate molar mass | 74.55 g/mol | 147.47 g/mol phase | solid (at STP) | solid (at STP) melting point | 770 °C | 310 °C boiling point | 1420 °C | 578 °C density | 1.98 g/cm^3 | 3.11 g/cm^3 solubility in water | soluble |
Units

Thermodynamic properties

| potassium chloride | rubidium nitrate molar heat of fusion | 26.28 kJ/mol (kilojoules per mole) | 4.6 kJ/mol (kilojoules per mole) specific heat of fusion | 0.3525 kJ/g (kilojoules per gram) | 0.031 kJ/g (kilojoules per gram)
Solid properties (at STP)

| potassium chloride | rubidium nitrate density | 1.98 g/cm^3 | 3.11 g/cm^3 refractive index | | 1.524
Units
