Input interpretation
gold(I) chloride
Chemical names and formulas

formula | AuCl name | gold(I) chloride IUPAC name | gold(+1) cation chloride alternate names | gold(+1) cation chloride mass fractions | Au (gold) 84.7% | Cl (chlorine) 15.3%
Structure diagram
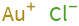
Structure diagram
Basic properties
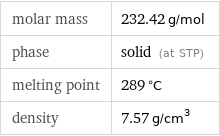
molar mass | 232.42 g/mol phase | solid (at STP) melting point | 289 °C density | 7.57 g/cm^3
Units

Solid properties (at STP)

density | 7.57 g/cm^3
Units

Thermodynamic properties

specific heat of formation Δ_fH° | solid | -0.1493 kJ/g molar heat of formation Δ_fH° | solid | -34.7 kJ/mol (at STP)
Chemical identifiers
![CAS number | 10294-29-8 PubChem CID number | 5460531 PubChem SID number | 24871740 SMILES identifier | [Cl-].[Au+] InChI identifier | InChI=1/Au.ClH/h;1H/q+1;/p-1/fAu.Cl/h;1h/qm;-1 MDL number | MFCD00046175](../image_source/ed54c3d0e0f73adf34b1ca5a9dc0a40d.png)
CAS number | 10294-29-8 PubChem CID number | 5460531 PubChem SID number | 24871740 SMILES identifier | [Cl-].[Au+] InChI identifier | InChI=1/Au.ClH/h;1H/q+1;/p-1/fAu.Cl/h;1h/qm;-1 MDL number | MFCD00046175
Ion equivalents

Au^+ (gold(I) cation) | 1 Cl^- (chloride anion) | 1