Input interpretation
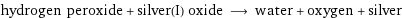
hydrogen peroxide + silver(I) oxide ⟶ water + oxygen + silver
Balanced equation

Balance the chemical equation algebraically: + ⟶ + + Add stoichiometric coefficients, c_i, to the reactants and products: c_1 + c_2 ⟶ c_3 + c_4 + c_5 Set the number of atoms in the reactants equal to the number of atoms in the products for H, O and Ag: H: | 2 c_1 = 2 c_3 O: | 2 c_1 + c_2 = c_3 + 2 c_4 Ag: | 2 c_2 = c_5 Since the coefficients are relative quantities and underdetermined, choose a coefficient to set arbitrarily. To keep the coefficients small, the arbitrary value is ordinarily one. For instance, set c_1 = 1 and solve the system of equations for the remaining coefficients: c_1 = 1 c_3 = 1 c_4 = c_2/2 + 1/2 c_5 = 2 c_2 The resulting system of equations is still underdetermined, so an additional coefficient must be set arbitrarily. Set c_2 = 1 and solve for the remaining coefficients: c_1 = 1 c_2 = 1 c_3 = 1 c_4 = 1 c_5 = 2 Substitute the coefficients into the chemical reaction to obtain the balanced equation: Answer: | | + ⟶ + + 2
Structures

+ ⟶ + +
Names
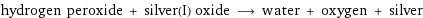
hydrogen peroxide + silver(I) oxide ⟶ water + oxygen + silver
Equilibrium constant
![K_c = ([H2O] [O2] [Ag]^2)/([H2O2] [Ag2O])](../image_source/7fab839e5762d922a609e0f021d177af.png)
K_c = ([H2O] [O2] [Ag]^2)/([H2O2] [Ag2O])
Rate of reaction
![rate = -(Δ[H2O2])/(Δt) = -(Δ[Ag2O])/(Δt) = (Δ[H2O])/(Δt) = (Δ[O2])/(Δt) = 1/2 (Δ[Ag])/(Δt) (assuming constant volume and no accumulation of intermediates or side products)](../image_source/ae932c48fd06015ad69a9d835868e517.png)
rate = -(Δ[H2O2])/(Δt) = -(Δ[Ag2O])/(Δt) = (Δ[H2O])/(Δt) = (Δ[O2])/(Δt) = 1/2 (Δ[Ag])/(Δt) (assuming constant volume and no accumulation of intermediates or side products)
Chemical names and formulas
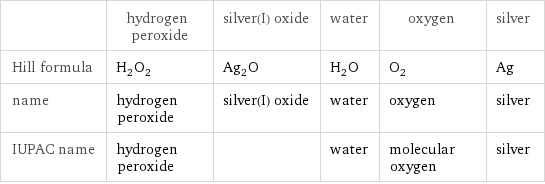
| hydrogen peroxide | silver(I) oxide | water | oxygen | silver Hill formula | H_2O_2 | Ag_2O | H_2O | O_2 | Ag name | hydrogen peroxide | silver(I) oxide | water | oxygen | silver IUPAC name | hydrogen peroxide | | water | molecular oxygen | silver