Input interpretation
sodium periodate
Chemical names and formulas

formula | NaIO_4 Hill formula | INaO_4 name | sodium periodate alternate names | metaperiodate | sodium (meta)periodate | sodium metaperiodate | sodium metaperiodate(sodium periodate) | sodium m-periodate mass fractions | I (iodine) 59.3% | Na (sodium) 10.7% | O (oxygen) 29.9%
Structure diagram
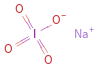
Structure diagram
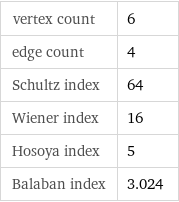
vertex count | 6 edge count | 4 Schultz index | 64 Wiener index | 16 Hosoya index | 5 Balaban index | 3.024
Basic properties
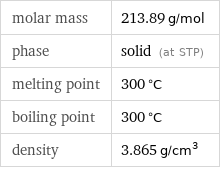
molar mass | 213.89 g/mol phase | solid (at STP) melting point | 300 °C boiling point | 300 °C density | 3.865 g/cm^3
Units

Solid properties (at STP)

density | 3.865 g/cm^3
Units

Thermodynamic properties

specific free energy of formation Δ_fG° | solid | -1.51 kJ/g molar free energy of formation Δ_fG° | solid | -323 kJ/mol specific heat of formation Δ_fH° | solid | -2.007 kJ/g molar heat of formation Δ_fH° | solid | -429.3 kJ/mol (at STP)
Chemical identifiers
![CAS number | 7790-28-5 PubChem CID number | 23667635 SMILES identifier | [O-]I(=O)(=O)=O.[Na+] InChI identifier | InChI=1/HIO4.Na/c2-1(3, 4)5;/h(H, 2, 3, 4, 5);/q;+1/p-1/fIO4.Na/q-1;m RTECS number | SD4550000 MDL number | MFCD00003534](../image_source/c0e222136065331867b4610f41980a55.png)
CAS number | 7790-28-5 PubChem CID number | 23667635 SMILES identifier | [O-]I(=O)(=O)=O.[Na+] InChI identifier | InChI=1/HIO4.Na/c2-1(3, 4)5;/h(H, 2, 3, 4, 5);/q;+1/p-1/fIO4.Na/q-1;m RTECS number | SD4550000 MDL number | MFCD00003534
NFPA label

NFPA label

NFPA health rating | 2 NFPA fire rating | 0 NFPA reactivity rating | 0
Toxicity properties

RTECS classes | other
Ion equivalents

Na^+ (sodium cation) | 1 (IO_4)^- (periodate anion) | 1