Input interpretation

polar aprotic solvents | molecular mass
Summary
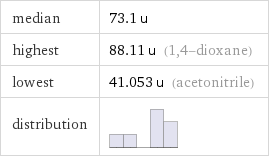
median | 73.1 u highest | 88.11 u (1, 4-dioxane) lowest | 41.053 u (acetonitrile) distribution |
Units

Ranked values
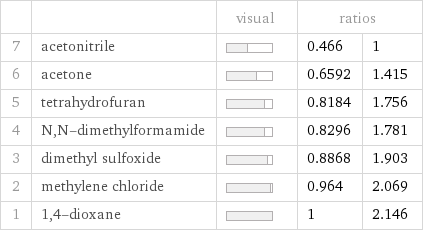
| | visual | ratios | 7 | acetonitrile | | 0.466 | 1 6 | acetone | | 0.6592 | 1.415 5 | tetrahydrofuran | | 0.8184 | 1.756 4 | N, N-dimethylformamide | | 0.8296 | 1.781 3 | dimethyl sulfoxide | | 0.8868 | 1.903 2 | methylene chloride | | 0.964 | 2.069 1 | 1, 4-dioxane | | 1 | 2.146
Molecular mass rankings
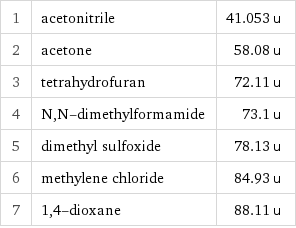
1 | acetonitrile | 41.053 u 2 | acetone | 58.08 u 3 | tetrahydrofuran | 72.11 u 4 | N, N-dimethylformamide | 73.1 u 5 | dimethyl sulfoxide | 78.13 u 6 | methylene chloride | 84.93 u 7 | 1, 4-dioxane | 88.11 u
Unit conversions for median molecular mass 73.1 u
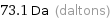
73.1 Da (daltons)
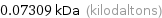
0.07309 kDa (kilodaltons)
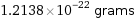
1.2138×10^-22 grams
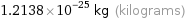
1.2138×10^-25 kg (kilograms)
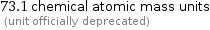
73.1 chemical atomic mass units (unit officially deprecated)

73.12 physical atomic mass units (unit officially deprecated)
Comparisons for median molecular mass 73.1 u
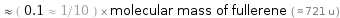
≈ ( 0.1 ≈ 1/10 ) × molecular mass of fullerene ( ≈ 721 u )
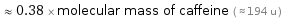
≈ 0.38 × molecular mass of caffeine ( ≈ 194 u )
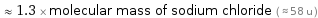
≈ 1.3 × molecular mass of sodium chloride ( ≈ 58 u )
Corresponding quantities
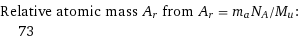
Relative atomic mass A_r from A_r = m_aN_A/M_u: | 73
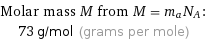
Molar mass M from M = m_aN_A: | 73 g/mol (grams per mole)
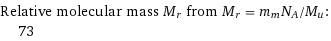
Relative molecular mass M_r from M_r = m_mN_A/M_u: | 73
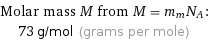
Molar mass M from M = m_mN_A: | 73 g/mol (grams per mole)