Input interpretation
lithium fluoride | 3, 3, 4, 4, 5, 5, 6, 6, 7, 7, 8, 8, 9, 9, 10, 10, 11, 11, 12, 12, 12-heneicosafluorododecyl acrylate
Chemical names and formulas
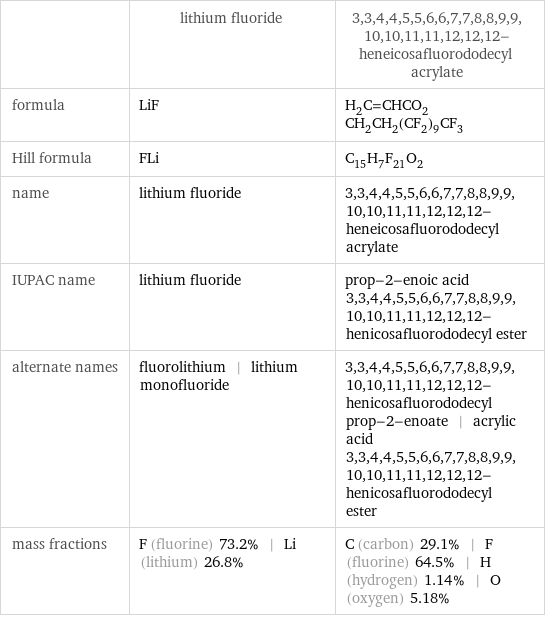
| lithium fluoride | 3, 3, 4, 4, 5, 5, 6, 6, 7, 7, 8, 8, 9, 9, 10, 10, 11, 11, 12, 12, 12-heneicosafluorododecyl acrylate formula | LiF | H_2C=CHCO_2CH_2CH_2(CF_2)_9CF_3 Hill formula | FLi | C_15H_7F_21O_2 name | lithium fluoride | 3, 3, 4, 4, 5, 5, 6, 6, 7, 7, 8, 8, 9, 9, 10, 10, 11, 11, 12, 12, 12-heneicosafluorododecyl acrylate IUPAC name | lithium fluoride | prop-2-enoic acid 3, 3, 4, 4, 5, 5, 6, 6, 7, 7, 8, 8, 9, 9, 10, 10, 11, 11, 12, 12, 12-henicosafluorododecyl ester alternate names | fluorolithium | lithium monofluoride | 3, 3, 4, 4, 5, 5, 6, 6, 7, 7, 8, 8, 9, 9, 10, 10, 11, 11, 12, 12, 12-henicosafluorododecyl prop-2-enoate | acrylic acid 3, 3, 4, 4, 5, 5, 6, 6, 7, 7, 8, 8, 9, 9, 10, 10, 11, 11, 12, 12, 12-henicosafluorododecyl ester mass fractions | F (fluorine) 73.2% | Li (lithium) 26.8% | C (carbon) 29.1% | F (fluorine) 64.5% | H (hydrogen) 1.14% | O (oxygen) 5.18%
Structure diagrams
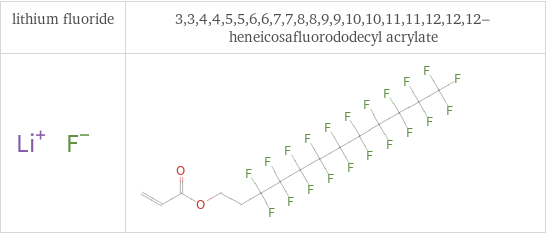
Structure diagrams
3D structure
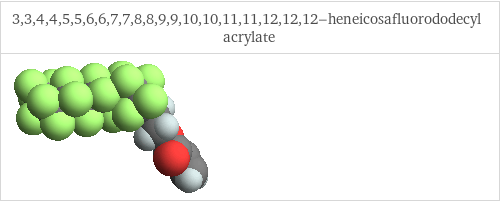
3D structure
Basic properties

| lithium fluoride | 3, 3, 4, 4, 5, 5, 6, 6, 7, 7, 8, 8, 9, 9, 10, 10, 11, 11, 12, 12, 12-heneicosafluorododecyl acrylate molar mass | 25.94 g/mol | 618.19 g/mol phase | solid (at STP) | solid (at STP) melting point | 845 °C | 50.5 °C boiling point | 1676 °C | 122 °C density | 2.64 g/cm^3 | 1.32 g/cm^3
Units

Thermodynamic properties
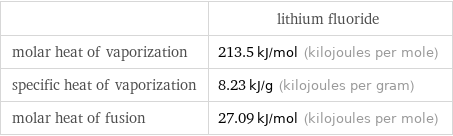
| lithium fluoride molar heat of vaporization | 213.5 kJ/mol (kilojoules per mole) specific heat of vaporization | 8.23 kJ/g (kilojoules per gram) molar heat of fusion | 27.09 kJ/mol (kilojoules per mole)
Solid properties (at STP)

| lithium fluoride | 3, 3, 4, 4, 5, 5, 6, 6, 7, 7, 8, 8, 9, 9, 10, 10, 11, 11, 12, 12, 12-heneicosafluorododecyl acrylate density | 2.64 g/cm^3 | 1.32 g/cm^3 vapor pressure | 0.07 mmHg | refractive index | 1.39937 |
Units
