Input interpretation
lithium iodide
Chemical names and formulas
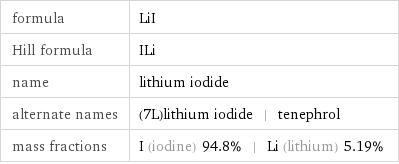
formula | LiI Hill formula | ILi name | lithium iodide alternate names | (7L)lithium iodide | tenephrol mass fractions | I (iodine) 94.8% | Li (lithium) 5.19%
Structure diagram

Structure diagram
Basic properties
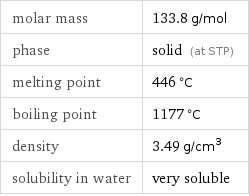
molar mass | 133.8 g/mol phase | solid (at STP) melting point | 446 °C boiling point | 1177 °C density | 3.49 g/cm^3 solubility in water | very soluble
Units

Solid properties (at STP)

density | 3.49 g/cm^3 vapor pressure | 112 mmHg (at 1000 °C)
Units

Thermodynamic properties

specific heat capacity c_p | solid | 0.381 J/(g K) molar heat capacity c_p | solid | 51 J/(mol K) specific free energy of formation Δ_fG° | solid | -2.02 kJ/g molar free energy of formation Δ_fG° | solid | -270.3 kJ/mol specific heat of formation Δ_fH° | solid | -2.02 kJ/g molar heat of formation Δ_fH° | solid | -270.4 kJ/mol molar heat of vaporization | 170.8 kJ/mol | specific heat of vaporization | 1.276 kJ/g | molar heat of fusion | 14.6 kJ/mol | specific heat of fusion | 0.109 kJ/g | (at STP)
Chemical identifiers
![CAS number | 10377-51-2 PubChem CID number | 66321 PubChem SID number | 24853051 SMILES identifier | [Li+].[I-] InChI identifier | InChI=1/HI.Li/h1H;/q;+1/p-1/fI.Li/h1h;/q-1;m MDL number | MFCD00011092](../image_source/892b79b2e65699548e42aa2ca3c11b6b.png)
CAS number | 10377-51-2 PubChem CID number | 66321 PubChem SID number | 24853051 SMILES identifier | [Li+].[I-] InChI identifier | InChI=1/HI.Li/h1H;/q;+1/p-1/fI.Li/h1h;/q-1;m MDL number | MFCD00011092
Ion equivalents

Li^+ (lithium cation) | 1 I^- (iodide anion) | 1