Input interpretation
cyclopentadienyl anion
Lewis structure
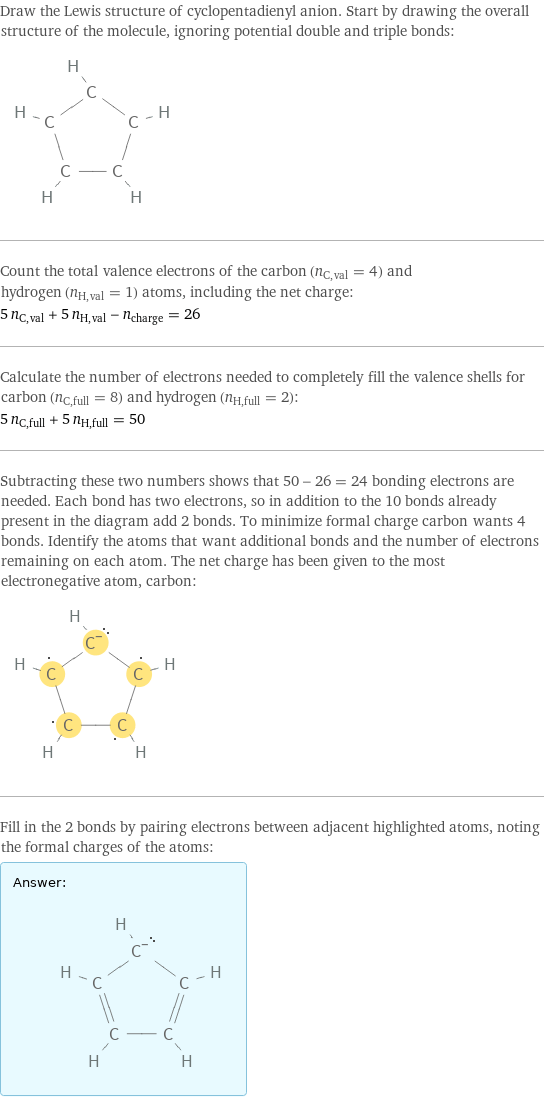
Draw the Lewis structure of cyclopentadienyl anion. Start by drawing the overall structure of the molecule, ignoring potential double and triple bonds: Count the total valence electrons of the carbon (n_C, val = 4) and hydrogen (n_H, val = 1) atoms, including the net charge: 5 n_C, val + 5 n_H, val - n_charge = 26 Calculate the number of electrons needed to completely fill the valence shells for carbon (n_C, full = 8) and hydrogen (n_H, full = 2): 5 n_C, full + 5 n_H, full = 50 Subtracting these two numbers shows that 50 - 26 = 24 bonding electrons are needed. Each bond has two electrons, so in addition to the 10 bonds already present in the diagram add 2 bonds. To minimize formal charge carbon wants 4 bonds. Identify the atoms that want additional bonds and the number of electrons remaining on each atom. The net charge has been given to the most electronegative atom, carbon: Fill in the 2 bonds by pairing electrons between adjacent highlighted atoms, noting the formal charges of the atoms: Answer: | |
General properties

formula | (C_5H_5)^- net ionic charge | -1 alternate names | Cp | cyclopenta-2, 4-dien-1-yl | cyclopentadienylide | cyclopentadienyl | cyclopentadienyl radical
Other properties
ion class | anions | cyclic pi donor | ionic ligands