Input interpretation
dichromate anion
Structure diagram

Structure diagram
General properties

formula | (Cr_2O_7)^(2-) net ionic charge | -2 alternate names | bichromate | dichromate | dichromate(2-)
Other properties

ion class | anions | chromium(VI) ions | oxoanions | polyatomic ions common sources of ion | sodium dichromate dihydrate (1 eq) | pyridinium dichromate (1 eq) | lithium dichromate hydrate (1 eq) | 4-carboxypyridinium dichromate (1 eq)
Thermodynamic properties
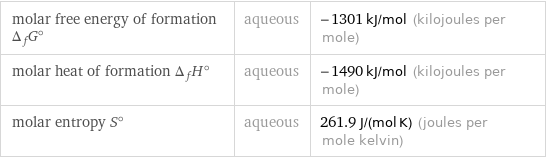
molar free energy of formation Δ_fG° | aqueous | -1301 kJ/mol (kilojoules per mole) molar heat of formation Δ_fH° | aqueous | -1490 kJ/mol (kilojoules per mole) molar entropy S° | aqueous | 261.9 J/(mol K) (joules per mole kelvin)