Input interpretation
lead(II) bromide
Chemical names and formulas

formula | PbBr_2 Hill formula | Br_2Pb name | lead(II) bromide alternate names | dibromolead | lead bromide | lead dibromide mass fractions | Br (bromine) 43.5% | Pb (lead) 56.5%
Structure diagram
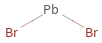
Structure diagram
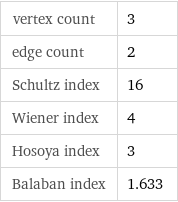
vertex count | 3 edge count | 2 Schultz index | 16 Wiener index | 4 Hosoya index | 3 Balaban index | 1.633
Basic properties
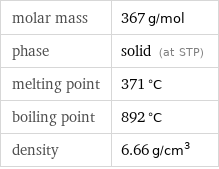
molar mass | 367 g/mol phase | solid (at STP) melting point | 371 °C boiling point | 892 °C density | 6.66 g/cm^3
Units

Solid properties (at STP)
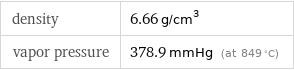
density | 6.66 g/cm^3 vapor pressure | 378.9 mmHg (at 849 °C)
Units

Thermodynamic properties
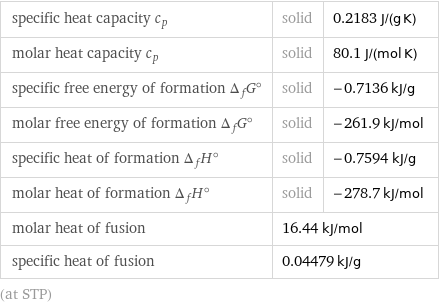
specific heat capacity c_p | solid | 0.2183 J/(g K) molar heat capacity c_p | solid | 80.1 J/(mol K) specific free energy of formation Δ_fG° | solid | -0.7136 kJ/g molar free energy of formation Δ_fG° | solid | -261.9 kJ/mol specific heat of formation Δ_fH° | solid | -0.7594 kJ/g molar heat of formation Δ_fH° | solid | -278.7 kJ/mol molar heat of fusion | 16.44 kJ/mol | specific heat of fusion | 0.04479 kJ/g | (at STP)
Chemical identifiers
![CAS number | 10031-22-8 SMILES identifier | Br[Pb]Br MDL number | MFCD00011156](../image_source/745dd2ce27e3c0b819f15a7f48ec1657.png)
CAS number | 10031-22-8 SMILES identifier | Br[Pb]Br MDL number | MFCD00011156