Input interpretation
lithium peroxide
Chemical names and formulas

formula | Li_2O_2 name | lithium peroxide IUPAC name | dilithium peroxide alternate names | dilithium peroxide mass fractions | Li (lithium) 30.3% | O (oxygen) 69.7%
Structure diagram

Structure diagram
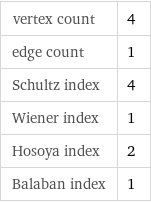
vertex count | 4 edge count | 1 Schultz index | 4 Wiener index | 1 Hosoya index | 2 Balaban index | 1
Basic properties
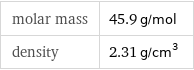
molar mass | 45.9 g/mol density | 2.31 g/cm^3
Units

Thermodynamic properties

specific heat of formation Δ_fH° | solid | -13.83 kJ/g molar heat of formation Δ_fH° | solid | -634.3 kJ/mol (at STP)
Chemical identifiers
![CAS number | 12031-80-0 PubChem CID number | 6093265 PubChem SID number | 24861460 SMILES identifier | [Li+].[Li+].[O-][O-] InChI identifier | InChI=1/2Li.O2/c;;1-2/q2*+1;-2 MDL number | MFCD00016184](../image_source/0f3f76800af6295a410c101ae6a05f5c.png)
CAS number | 12031-80-0 PubChem CID number | 6093265 PubChem SID number | 24861460 SMILES identifier | [Li+].[Li+].[O-][O-] InChI identifier | InChI=1/2Li.O2/c;;1-2/q2*+1;-2 MDL number | MFCD00016184
NFPA label

NFPA label

NFPA hazards | oxidizing agent
Ion equivalents
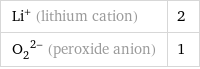
Li^+ (lithium cation) | 2 (O_2)^(2-) (peroxide anion) | 1