Input interpretation

van der Waals equation of state
Equation

(P + a/V_m^2) (V_m - b) = R T | P | pressure a | van der Waals constant a b | van der Waals constant b V_m | molar volume T | temperature R | molar gas constant (≈ 8.314 J/(mol K))
Units

Input values
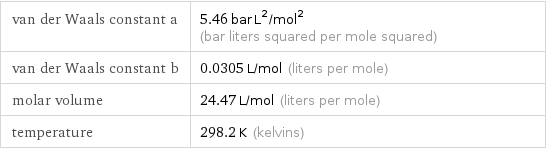
van der Waals constant a | 5.46 bar L^2/mol^2 (bar liters squared per mole squared) van der Waals constant b | 0.0305 L/mol (liters per mole) molar volume | 24.47 L/mol (liters per mole) temperature | 298.2 K (kelvins)
Result

pressure | 100.5 kPa (kilopascals) = 14.58 psi (pounds-force per square inch) = 2100 lbf/ft^2 (pounds-force per square foot) = 0.1005 MPa (megapascals) = 100536 Pa (pascals) = 0.9922 atm (atmospheres) = 1005 mbar (millibars) = 754.1 mmHg (millimeters of mercury)