Input interpretation
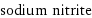
sodium nitrite
Chemical names and formulas
![formula | NaNO_2 Hill formula | NNaO_2 name | sodium nitrite alternate names | anti-rust | azotyn sodowy [Polish] | nitrous acid, sodium salt mass fractions | N (nitrogen) 20.3% | Na (sodium) 33.3% | O (oxygen) 46.4%](../image_source/18ef825424e2b7a279c3da43b3c02ca2.png)
formula | NaNO_2 Hill formula | NNaO_2 name | sodium nitrite alternate names | anti-rust | azotyn sodowy [Polish] | nitrous acid, sodium salt mass fractions | N (nitrogen) 20.3% | Na (sodium) 33.3% | O (oxygen) 46.4%
Structure diagram

Structure diagram
vertex count | 4 edge count | 2 Schultz index | 16 Wiener index | 4 Hosoya index | 3 Balaban index | 1.633
Basic properties
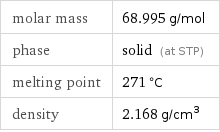
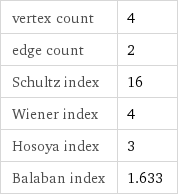
molar mass | 68.995 g/mol phase | solid (at STP) melting point | 271 °C density | 2.168 g/cm^3
Units

Solid properties (at STP)
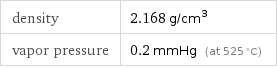
density | 2.168 g/cm^3 vapor pressure | 0.2 mmHg (at 525 °C)
Units

Thermodynamic properties

specific free energy of formation Δ_fG° | solid | -4.125 kJ/g molar free energy of formation Δ_fG° | solid | -284.6 kJ/mol specific heat of formation Δ_fH° | solid | -5.203 kJ/g molar heat of formation Δ_fH° | solid | -359 kJ/mol molar heat of fusion | 14.9 kJ/mol | specific heat of fusion | 0.216 kJ/g | (at STP)
Chemical identifiers
![CAS number | 7632-00-0 PubChem CID number | 23668193 PubChem SID number | 24867034 SMILES identifier | N(=O)[O-].[Na+] InChI identifier | InChI=1/HNO2.Na/c2-1-3;/h(H, 2, 3);/q;+1/p-1/fNO2.Na/q-1;m RTECS number | RA1225000 MDL number | MFCD00011118](../image_source/493bb86bffde0bd904b2c7c67006b91f.png)
CAS number | 7632-00-0 PubChem CID number | 23668193 PubChem SID number | 24867034 SMILES identifier | N(=O)[O-].[Na+] InChI identifier | InChI=1/HNO2.Na/c2-1-3;/h(H, 2, 3);/q;+1/p-1/fNO2.Na/q-1;m RTECS number | RA1225000 MDL number | MFCD00011118
NFPA label

NFPA label
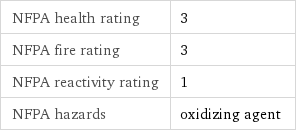
NFPA health rating | 3 NFPA fire rating | 3 NFPA reactivity rating | 1 NFPA hazards | oxidizing agent
Safety properties

autoignition point | 489 °C
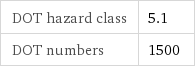
DOT hazard class | 5.1 DOT numbers | 1500
Toxicity properties
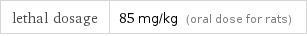
lethal dosage | 85 mg/kg (oral dose for rats)

probable lethal dose for man | 30 mL (milliliters) RTECS classes | tumorigen | mutagen | reproductive effector | human data | primary irritant
Ion equivalents

Na^+ (sodium cation) | 1 (NO_2)^- (nitrite anion) | 1