Input interpretation

diamagnetic elements | nuclear radius
Summary
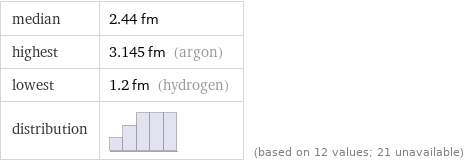
median | 2.44 fm highest | 3.145 fm (argon) lowest | 1.2 fm (hydrogen) distribution | | (based on 12 values; 21 unavailable)
Entities with missing values
antimony | arsenic | bismuth | bromine | cadmium | copper | gallium | germanium | gold | indium | ... (total: 21)
Distribution plots
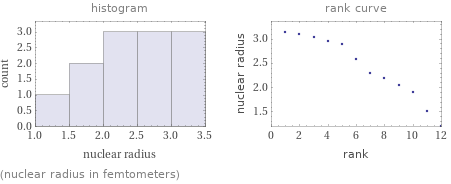
(nuclear radius in femtometers)
Nuclear radius rankings
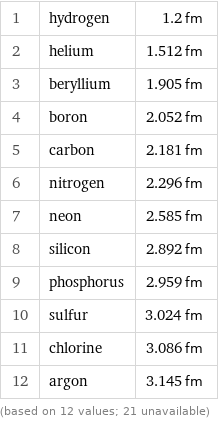
1 | hydrogen | 1.2 fm 2 | helium | 1.512 fm 3 | beryllium | 1.905 fm 4 | boron | 2.052 fm 5 | carbon | 2.181 fm 6 | nitrogen | 2.296 fm 7 | neon | 2.585 fm 8 | silicon | 2.892 fm 9 | phosphorus | 2.959 fm 10 | sulfur | 3.024 fm 11 | chlorine | 3.086 fm 12 | argon | 3.145 fm (based on 12 values; 21 unavailable)
Unit conversions for median nuclear radius 2.44 fm
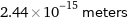
2.44×10^-15 meters
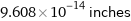
9.608×10^-14 inches
Comparisons for median nuclear radius 2.44 fm

≈ 0.87 × classical electron radius ( ≈ 2.8×10^-15 m )
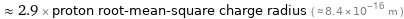
≈ 2.9 × proton root-mean-square charge radius ( ≈ 8.4×10^-16 m )