Input interpretation
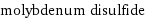
molybdenum disulfide
Chemical names and formulas

formula | MoS_2 name | molybdenum disulfide IUPAC name | dithioxomolybdenum alternate names | disulfanylidene molybdenum | dithioxomolybdenum | molybdenite | molybdenum(IV) sulfide | molybdenum sulfide | molykote mass fractions | Mo (molybdenum) 59.9% | S (sulfur) 40.1%
Structure diagram

Structure diagram
vertex count | 3 edge count | 2 Schultz index | 16 Wiener index | 4 Hosoya index | 3 Balaban index | 1.633
Basic properties

molar mass | 160.1 g/mol phase | solid (at STP) melting point | 2375 °C density | 5.06 g/cm^3 solubility in water | insoluble particle size | 2 µm
Solid properties (at STP)

density | 5.06 g/cm^3
Units

Thermodynamic properties
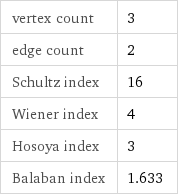
specific heat capacity c_p | solid | 3.973 J/(g K) molar heat capacity c_p | solid | 636 J/(mol K) specific free energy of formation Δ_fG° | solid | -1.411 kJ/g molar free energy of formation Δ_fG° | solid | -225.9 kJ/mol specific heat of formation Δ_fH° | solid | -1.469 kJ/g molar heat of formation Δ_fH° | solid | -235.1 kJ/mol (at STP)
Chemical identifiers
![CAS number | 1317-33-5 PubChem CID number | 14823 PubChem SID number | 24854060 SMILES identifier | S=[Mo]=S InChI identifier | InChI=1/Mo.2S/rMoS2/c2-1-3 RTECS number | QA4697000 MDL number | MFCD00003470](../image_source/8eb8ba3cf8483f403daf0043da12ed0e.png)
CAS number | 1317-33-5 PubChem CID number | 14823 PubChem SID number | 24854060 SMILES identifier | S=[Mo]=S InChI identifier | InChI=1/Mo.2S/rMoS2/c2-1-3 RTECS number | QA4697000 MDL number | MFCD00003470
Toxicity properties

RTECS classes | mutagen