Input interpretation
2 kg of acetone
Basic properties for 2 kg

mass | 2 kg (kilograms) 4.41 lb (pounds) 4 pounds 6.5 ounces 2000 grams molar amount | 34.4 mol (moles) volume | 2.53 L (liters) 0.00253 m^3 (cubic meters) 2530 cm^3 (cubic centimeters) 85.5 fl oz (fluid ounces) 5.34 pints 2.67 quarts 0.668 gallons 0.0893 ft^3 (cubic feet) 154 in^3 (cubic inches) (at STP)
Corresponding quantities
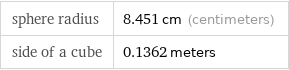
sphere radius | 8.451 cm (centimeters) side of a cube | 0.1362 meters
Thermodynamic properties for 2 kg

enthalpy of hydration | -1367 kJ (kilojoules) | heat capacity C_p | gaseous | 2565 J/K | liquid | 4349 J/K free energy of formation Δ_fG° | gaseous | -5258 kJ heat of formation Δ_fH° | gaseous | -7476 kJ latent heat of vaporization | 1100 kJ (kilojoules) | latent heat of fusion | 199 kJ (kilojoules) |
Units
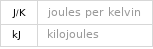
Energy vs. temperature for 2 kg
(relative to STP)
Units

Phase change energies for 2 kg from 25 °C

energy required to heat to boiling point | 135 kJ (kilojoules) energy required to convert to vapor | 1100 kJ (kilojoules) energy required to heat to boiling point and convert to vapor | 1230 kJ (kilojoules) energy released from cooling to freezing point | 518 kJ (kilojoules) energy released from converting to solid | 199 kJ (kilojoules) energy released from cooling to freezing point and converting to solid | 716 kJ (kilojoules)
Mass composition for 2 kg
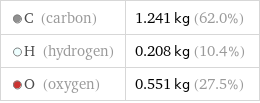
C (carbon) | 1.241 kg (62.0%) H (hydrogen) | 0.208 kg (10.4%) O (oxygen) | 0.551 kg (27.5%)
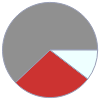
Mass composition for 2 kg
Lewis structure

Draw the Lewis structure of acetone. Start by drawing the overall structure of the molecule, ignoring potential double and triple bonds: Count the total valence electrons of the carbon (n_C, val = 4), hydrogen (n_H, val = 1), and oxygen (n_O, val = 6) atoms: 3 n_C, val + 6 n_H, val + n_O, val = 24 Calculate the number of electrons needed to completely fill the valence shells for carbon (n_C, full = 8), hydrogen (n_H, full = 2), and oxygen (n_O, full = 8): 3 n_C, full + 6 n_H, full + n_O, full = 44 Subtracting these two numbers shows that 44 - 24 = 20 bonding electrons are needed. Each bond has two electrons, so in addition to the 9 bonds already present in the diagram add 1 bond. To minimize formal charge oxygen wants 2 bonds and carbon wants 4 bonds. Identify the atoms that want additional bonds and the number of electrons remaining on each atom: Fill in the 1 bond by pairing electrons between adjacent highlighted atoms: Answer: | |
Chemical names and formulas
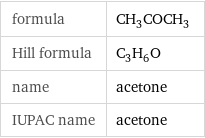
formula | CH_3COCH_3 Hill formula | C_3H_6O name | acetone IUPAC name | acetone