Input interpretation

hydrogen chromate anion | ion class
Result
anions | chromium(VI) ions | oxoanions | polyatomic ions
Ionic radii
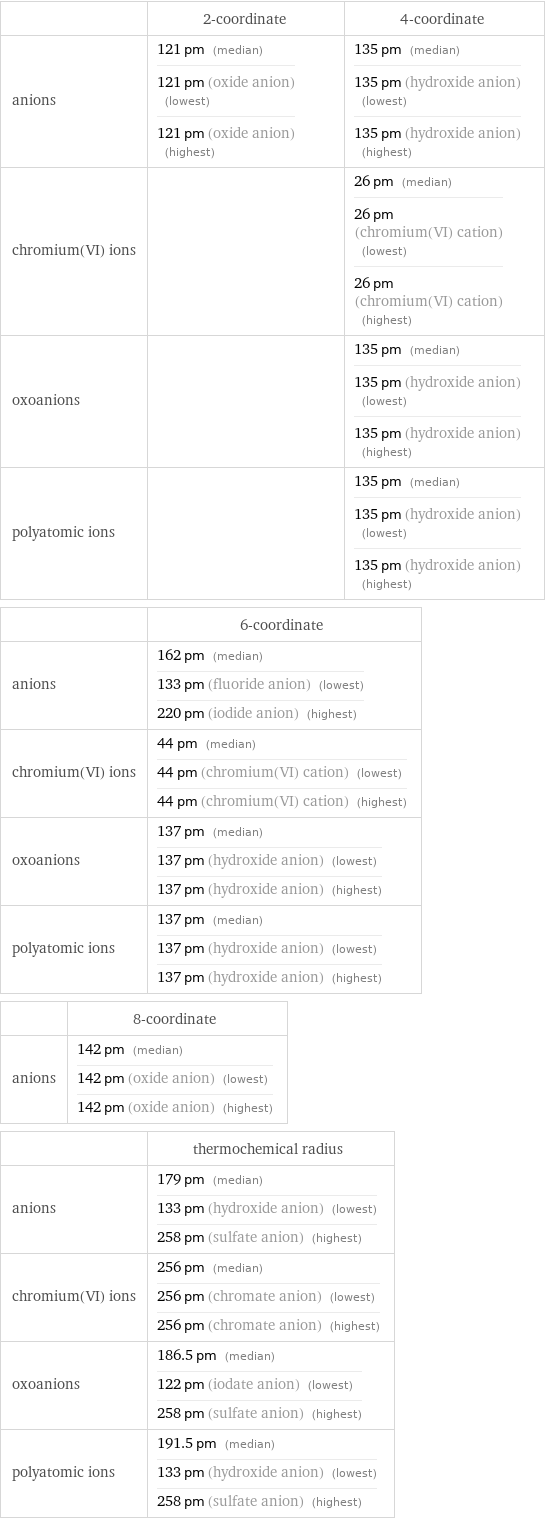
| 2-coordinate | 4-coordinate anions | 121 pm (median) 121 pm (oxide anion) (lowest) 121 pm (oxide anion) (highest) | 135 pm (median) 135 pm (hydroxide anion) (lowest) 135 pm (hydroxide anion) (highest) chromium(VI) ions | | 26 pm (median) 26 pm (chromium(VI) cation) (lowest) 26 pm (chromium(VI) cation) (highest) oxoanions | | 135 pm (median) 135 pm (hydroxide anion) (lowest) 135 pm (hydroxide anion) (highest) polyatomic ions | | 135 pm (median) 135 pm (hydroxide anion) (lowest) 135 pm (hydroxide anion) (highest) | 6-coordinate anions | 162 pm (median) 133 pm (fluoride anion) (lowest) 220 pm (iodide anion) (highest) chromium(VI) ions | 44 pm (median) 44 pm (chromium(VI) cation) (lowest) 44 pm (chromium(VI) cation) (highest) oxoanions | 137 pm (median) 137 pm (hydroxide anion) (lowest) 137 pm (hydroxide anion) (highest) polyatomic ions | 137 pm (median) 137 pm (hydroxide anion) (lowest) 137 pm (hydroxide anion) (highest) | 8-coordinate anions | 142 pm (median) 142 pm (oxide anion) (lowest) 142 pm (oxide anion) (highest) | thermochemical radius anions | 179 pm (median) 133 pm (hydroxide anion) (lowest) 258 pm (sulfate anion) (highest) chromium(VI) ions | 256 pm (median) 256 pm (chromate anion) (lowest) 256 pm (chromate anion) (highest) oxoanions | 186.5 pm (median) 122 pm (iodate anion) (lowest) 258 pm (sulfate anion) (highest) polyatomic ions | 191.5 pm (median) 133 pm (hydroxide anion) (lowest) 258 pm (sulfate anion) (highest)