Input interpretation

ionic diatomics | electron count
Summary
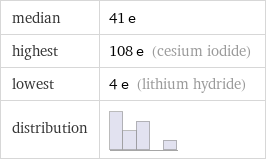
median | 41 e highest | 108 e (cesium iodide) lowest | 4 e (lithium hydride) distribution |
Units

Electron count rankings
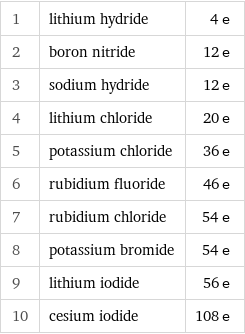
1 | lithium hydride | 4 e 2 | boron nitride | 12 e 3 | sodium hydride | 12 e 4 | lithium chloride | 20 e 5 | potassium chloride | 36 e 6 | rubidium fluoride | 46 e 7 | rubidium chloride | 54 e 8 | potassium bromide | 54 e 9 | lithium iodide | 56 e 10 | cesium iodide | 108 e
Corresponding quantity
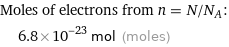
Moles of electrons from n = N/N_A: | 6.8×10^-23 mol (moles)