Input interpretation
magnesium perchlorate | sodium bromate
Chemical names and formulas
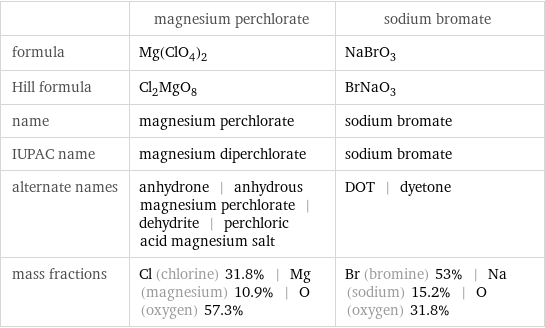
| magnesium perchlorate | sodium bromate formula | Mg(ClO_4)_2 | NaBrO_3 Hill formula | Cl_2MgO_8 | BrNaO_3 name | magnesium perchlorate | sodium bromate IUPAC name | magnesium diperchlorate | sodium bromate alternate names | anhydrone | anhydrous magnesium perchlorate | dehydrite | perchloric acid magnesium salt | DOT | dyetone mass fractions | Cl (chlorine) 31.8% | Mg (magnesium) 10.9% | O (oxygen) 57.3% | Br (bromine) 53% | Na (sodium) 15.2% | O (oxygen) 31.8%
Structure diagrams
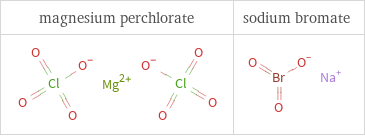
Structure diagrams
Basic properties
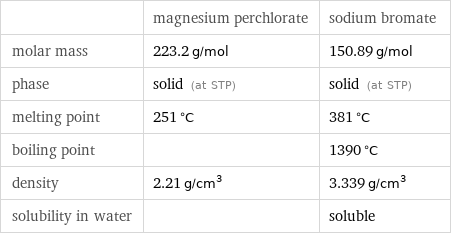
| magnesium perchlorate | sodium bromate molar mass | 223.2 g/mol | 150.89 g/mol phase | solid (at STP) | solid (at STP) melting point | 251 °C | 381 °C boiling point | | 1390 °C density | 2.21 g/cm^3 | 3.339 g/cm^3 solubility in water | | soluble
Units

Solid properties (at STP)

| magnesium perchlorate | sodium bromate density | 2.21 g/cm^3 | 3.339 g/cm^3
Units
