Input interpretation

exploding massive star elements | atomic diameter
Summary
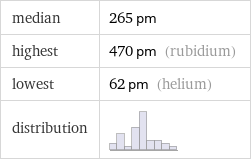
median | 265 pm highest | 470 pm (rubidium) lowest | 62 pm (helium) distribution |
Units

Plot
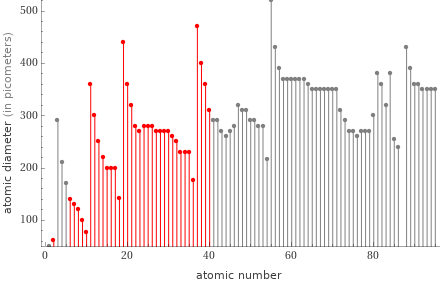
Plot
Periodic table location

Periodic table location
Atomic radii properties
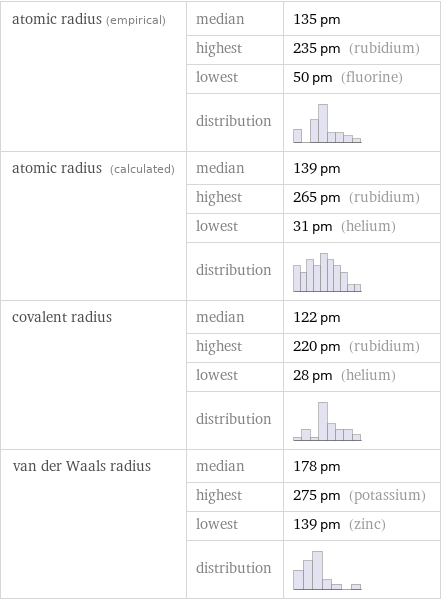
atomic radius (empirical) | median | 135 pm | highest | 235 pm (rubidium) | lowest | 50 pm (fluorine) | distribution | atomic radius (calculated) | median | 139 pm | highest | 265 pm (rubidium) | lowest | 31 pm (helium) | distribution | covalent radius | median | 122 pm | highest | 220 pm (rubidium) | lowest | 28 pm (helium) | distribution | van der Waals radius | median | 178 pm | highest | 275 pm (potassium) | lowest | 139 pm (zinc) | distribution |
Units

Distribution plots
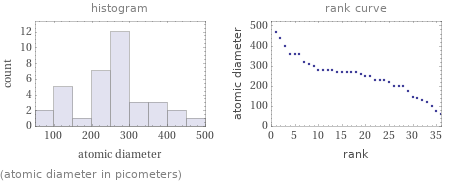
(atomic diameter in picometers)
Atomic diameter rankings
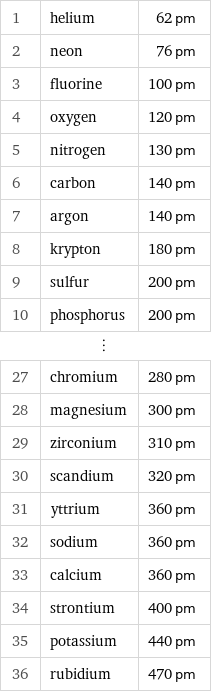
1 | helium | 62 pm 2 | neon | 76 pm 3 | fluorine | 100 pm 4 | oxygen | 120 pm 5 | nitrogen | 130 pm 6 | carbon | 140 pm 7 | argon | 140 pm 8 | krypton | 180 pm 9 | sulfur | 200 pm 10 | phosphorus | 200 pm ⋮ | | 27 | chromium | 280 pm 28 | magnesium | 300 pm 29 | zirconium | 310 pm 30 | scandium | 320 pm 31 | yttrium | 360 pm 32 | sodium | 360 pm 33 | calcium | 360 pm 34 | strontium | 400 pm 35 | potassium | 440 pm 36 | rubidium | 470 pm
Unit conversions for median atomic diameter 265 pm
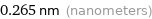
0.265 nm (nanometers)
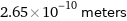
2.65×10^-10 meters
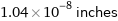
1.04×10^-8 inches
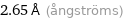
2.65 Å (ångströms)
Comparisons for median atomic diameter 265 pm
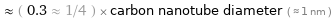
≈ ( 0.3 ≈ 1/4 ) × carbon nanotube diameter ( ≈ 1 nm )
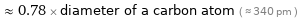
≈ 0.78 × diameter of a carbon atom ( ≈ 340 pm )
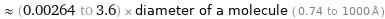
≈ (0.00264 to 3.6) × diameter of a molecule ( 0.74 to 1000 Å )