Input interpretation
magnesium phosphate | nickel(II) stannate dihydrate
Chemical names and formulas
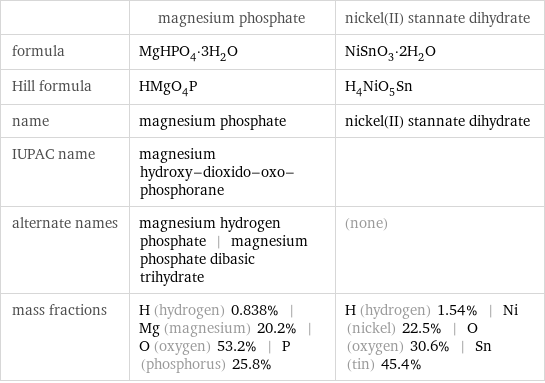
| magnesium phosphate | nickel(II) stannate dihydrate formula | MgHPO_4·3H_2O | NiSnO_3·2H_2O Hill formula | HMgO_4P | H_4NiO_5Sn name | magnesium phosphate | nickel(II) stannate dihydrate IUPAC name | magnesium hydroxy-dioxido-oxo-phosphorane | alternate names | magnesium hydrogen phosphate | magnesium phosphate dibasic trihydrate | (none) mass fractions | H (hydrogen) 0.838% | Mg (magnesium) 20.2% | O (oxygen) 53.2% | P (phosphorus) 25.8% | H (hydrogen) 1.54% | Ni (nickel) 22.5% | O (oxygen) 30.6% | Sn (tin) 45.4%
Structure diagrams
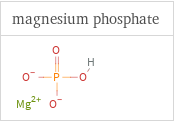
Structure diagrams
Basic properties
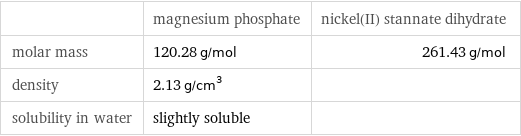
| magnesium phosphate | nickel(II) stannate dihydrate molar mass | 120.28 g/mol | 261.43 g/mol density | 2.13 g/cm^3 | solubility in water | slightly soluble |
Units
