Input interpretation
mercury(I) carbonate | lead arsenite
Chemical names and formulas
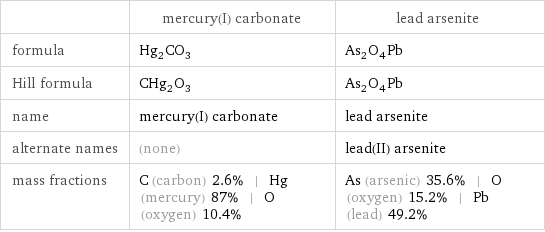
| mercury(I) carbonate | lead arsenite formula | Hg_2CO_3 | As_2O_4Pb Hill formula | CHg_2O_3 | As_2O_4Pb name | mercury(I) carbonate | lead arsenite alternate names | (none) | lead(II) arsenite mass fractions | C (carbon) 2.6% | Hg (mercury) 87% | O (oxygen) 10.4% | As (arsenic) 35.6% | O (oxygen) 15.2% | Pb (lead) 49.2%
Structure diagrams
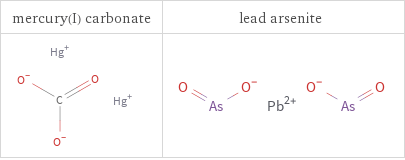
Structure diagrams
Basic properties

| mercury(I) carbonate | lead arsenite molar mass | 461.19 g/mol | 421 g/mol density | | 5.85 g/cm^3 solubility in water | | insoluble
Units
