Input interpretation

polar solvents | molar heat of fusion
Summary
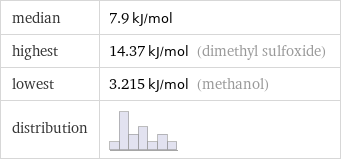
median | 7.9 kJ/mol highest | 14.37 kJ/mol (dimethyl sulfoxide) lowest | 3.215 kJ/mol (methanol) distribution |
Units

Distribution plots
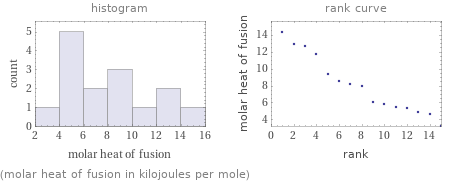
(molar heat of fusion in kilojoules per mole)
Molar heat of fusion rankings
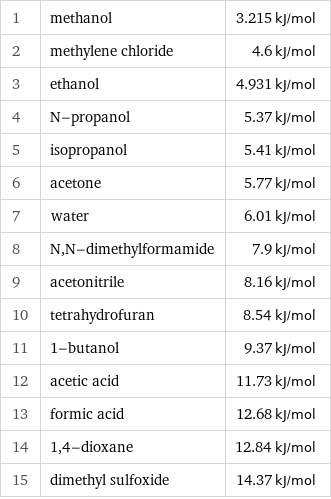
1 | methanol | 3.215 kJ/mol 2 | methylene chloride | 4.6 kJ/mol 3 | ethanol | 4.931 kJ/mol 4 | N-propanol | 5.37 kJ/mol 5 | isopropanol | 5.41 kJ/mol 6 | acetone | 5.77 kJ/mol 7 | water | 6.01 kJ/mol 8 | N, N-dimethylformamide | 7.9 kJ/mol 9 | acetonitrile | 8.16 kJ/mol 10 | tetrahydrofuran | 8.54 kJ/mol 11 | 1-butanol | 9.37 kJ/mol 12 | acetic acid | 11.73 kJ/mol 13 | formic acid | 12.68 kJ/mol 14 | 1, 4-dioxane | 12.84 kJ/mol 15 | dimethyl sulfoxide | 14.37 kJ/mol
Unit conversions for median molar heat of fusion 7.9 kJ/mol
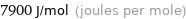
7900 J/mol (joules per mole)
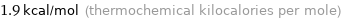
1.9 kcal/mol (thermochemical kilocalories per mole)
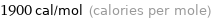
1900 cal/mol (calories per mole)

0.082 eV (molar electronvolts)