Input interpretation

elements | atomic radius
Summary
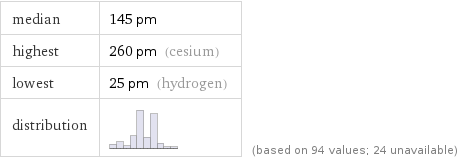
median | 145 pm highest | 260 pm (cesium) lowest | 25 pm (hydrogen) distribution | | (based on 94 values; 24 unavailable)
Entities with missing values

francium | curium | berkelium | californium | einsteinium | fermium | mendelevium | nobelium | lawrencium | rutherfordium | ... (total: 24)
Plot
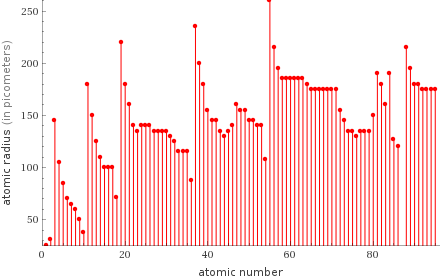
Plot
Periodic table location
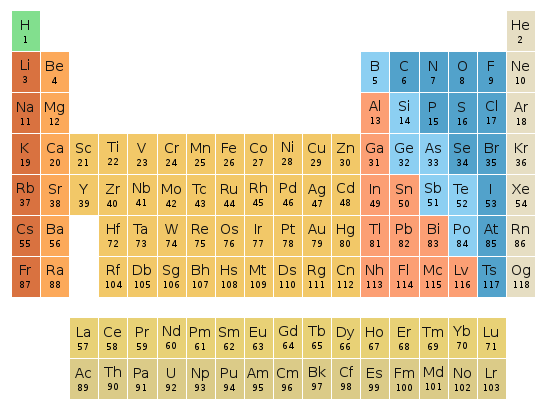
Periodic table location
Atomic radii properties
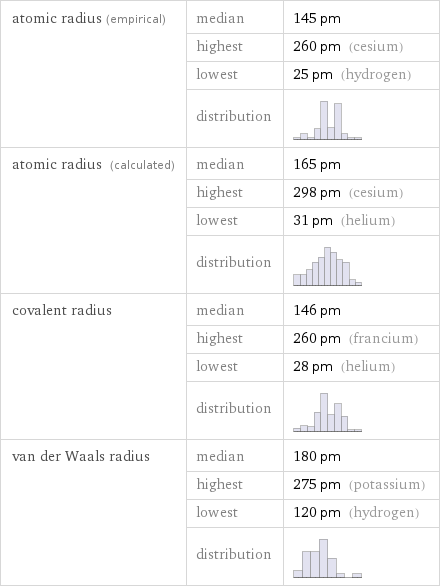
atomic radius (empirical) | median | 145 pm | highest | 260 pm (cesium) | lowest | 25 pm (hydrogen) | distribution | atomic radius (calculated) | median | 165 pm | highest | 298 pm (cesium) | lowest | 31 pm (helium) | distribution | covalent radius | median | 146 pm | highest | 260 pm (francium) | lowest | 28 pm (helium) | distribution | van der Waals radius | median | 180 pm | highest | 275 pm (potassium) | lowest | 120 pm (hydrogen) | distribution |
Units

Distribution plots
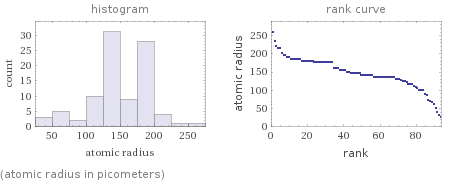
(atomic radius in picometers)
Atomic radius rankings
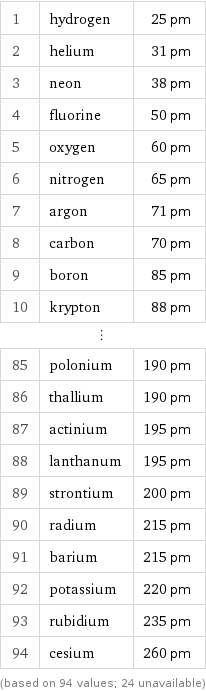
1 | hydrogen | 25 pm 2 | helium | 31 pm 3 | neon | 38 pm 4 | fluorine | 50 pm 5 | oxygen | 60 pm 6 | nitrogen | 65 pm 7 | argon | 71 pm 8 | carbon | 70 pm 9 | boron | 85 pm 10 | krypton | 88 pm ⋮ | | 85 | polonium | 190 pm 86 | thallium | 190 pm 87 | actinium | 195 pm 88 | lanthanum | 195 pm 89 | strontium | 200 pm 90 | radium | 215 pm 91 | barium | 215 pm 92 | potassium | 220 pm 93 | rubidium | 235 pm 94 | cesium | 260 pm (based on 94 values; 24 unavailable)
Unit conversions for median atomic radius 145 pm
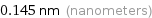
0.145 nm (nanometers)
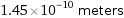
1.45×10^-10 meters
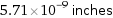
5.71×10^-9 inches
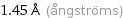
1.45 Å (ångströms)
Comparisons for median atomic radius 145 pm
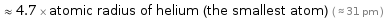
≈ 4.7 × atomic radius of helium (the smallest atom) ( ≈ 31 pm )
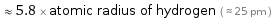
≈ 5.8 × atomic radius of hydrogen ( ≈ 25 pm )