Input interpretation

alkaline earth metals | melting point
Summary

median | 752 °C highest | 1287 °C (beryllium) lowest | 650 °C (magnesium) distribution |
Plot
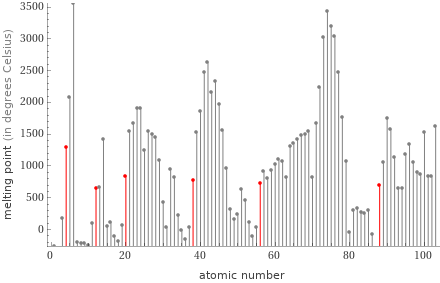
Plot
Melting point rankings

1 | magnesium | 650 °C 2 | radium | 700 °C 3 | barium | 727 °C 4 | strontium | 777 °C 5 | calcium | 842 °C 6 | beryllium | 1287 °C
Unit conversions for median melting point 752 °C
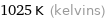
1025 K (kelvins)

1390 °F (degrees Fahrenheit)
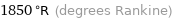
1850 °R (degrees Rankine)

602 °Ré (degrees Réaumur)
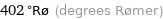
402 °Rø (degrees Rømer)
Comparison for median melting point 752 °C

548 °C below to 52 °C above typical temperature of magma (700 to 1300 °C)
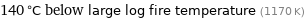
140 °C below large log fire temperature (1170 K)
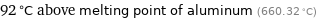
92 °C above melting point of aluminum (660.32 °C)
Blackbody information for median melting point 752 °C (degrees Celsius)
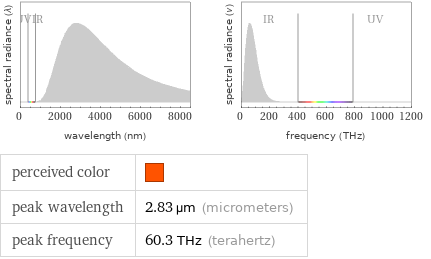
perceived color | peak wavelength | 2.83 µm (micrometers) peak frequency | 60.3 THz (terahertz)
Corresponding quantities

Thermodynamic energy E from E = kT: | 88 meV (millielectronvolts)
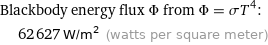
Blackbody energy flux Φ from Φ = σT^4: | 62627 W/m^2 (watts per square meter)
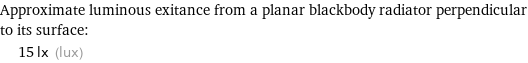
Approximate luminous exitance from a planar blackbody radiator perpendicular to its surface: | 15 lx (lux)
Thermodynamic properties
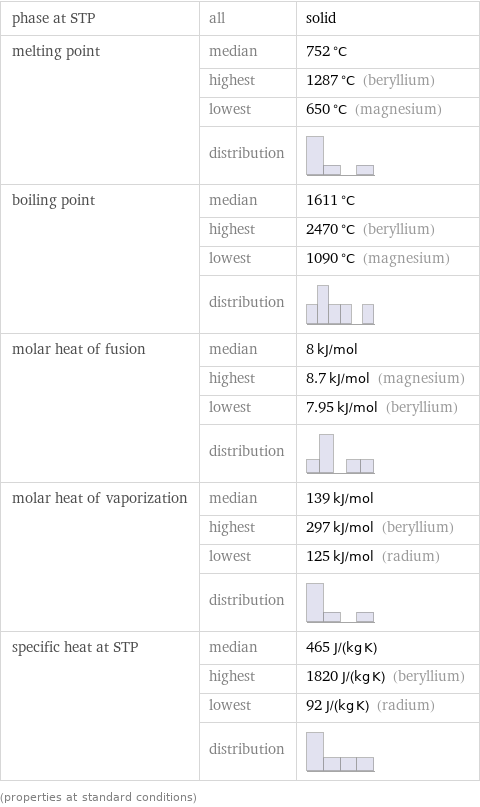
phase at STP | all | solid melting point | median | 752 °C | highest | 1287 °C (beryllium) | lowest | 650 °C (magnesium) | distribution | boiling point | median | 1611 °C | highest | 2470 °C (beryllium) | lowest | 1090 °C (magnesium) | distribution | molar heat of fusion | median | 8 kJ/mol | highest | 8.7 kJ/mol (magnesium) | lowest | 7.95 kJ/mol (beryllium) | distribution | molar heat of vaporization | median | 139 kJ/mol | highest | 297 kJ/mol (beryllium) | lowest | 125 kJ/mol (radium) | distribution | specific heat at STP | median | 465 J/(kg K) | highest | 1820 J/(kg K) (beryllium) | lowest | 92 J/(kg K) (radium) | distribution | (properties at standard conditions)