Input interpretation

Brønsted-Lowry bases | molar heat of fusion
Summary

median | 7.6 kJ/mol highest | 26 kJ/mol (sodium hydride) lowest | 5.66 kJ/mol (ammonia) | (based on 5 values; 5 unavailable)
Entities with missing values
butyllithium | methylmagnesium chloride | ethylmagnesium chloride | cyclohexene, methyl | phenylmagnesium bromide (total: 5)
Molar heat of fusion rankings

1 | ammonia | 5.66 kJ/mol 2 | water | 6.01 kJ/mol 3 | 2-methyl-2-butene | 7.6 kJ/mol 4 | trimethylamine | 7 kJ/mol 5 | sodium hydride | 26 kJ/mol (based on 5 values; 5 unavailable)
Unit conversions for median molar heat of fusion 7.6 kJ/mol
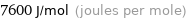
7600 J/mol (joules per mole)
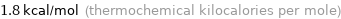
1.8 kcal/mol (thermochemical kilocalories per mole)
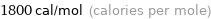
1800 cal/mol (calories per mole)

0.079 eV (molar electronvolts)