Input interpretation

polyatomic ions | formula
Table
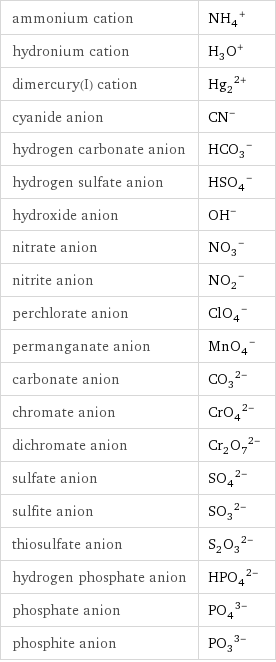
ammonium cation | (NH_4)^+ hydronium cation | (H_3O)^+ dimercury(I) cation | (Hg_2)^(2+) cyanide anion | (CN)^- hydrogen carbonate anion | (HCO_3)^- hydrogen sulfate anion | (HSO_4)^- hydroxide anion | (OH)^- nitrate anion | (NO_3)^- nitrite anion | (NO_2)^- perchlorate anion | (ClO_4)^- permanganate anion | (MnO_4)^- carbonate anion | (CO_3)^(2-) chromate anion | (CrO_4)^(2-) dichromate anion | (Cr_2O_7)^(2-) sulfate anion | (SO_4)^(2-) sulfite anion | (SO_3)^(2-) thiosulfate anion | (S_2O_3)^(2-) hydrogen phosphate anion | (HPO_4)^(2-) phosphate anion | (PO_4)^(3-) phosphite anion | (PO_3)^(3-)