Input interpretation
lead(II) nitrate
Chemical names and formulas

formula | Pb(NO_3)_2 Hill formula | N_2O_6Pb name | lead(II) nitrate IUPAC name | plumbous dinitrate alternate names | lead(+2) cation dinitrate | plumbous dinitrate mass fractions | N (nitrogen) 8.46% | O (oxygen) 29% | Pb (lead) 62.6%
Structure diagram
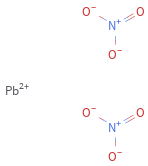
Structure diagram
Basic properties
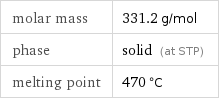
molar mass | 331.2 g/mol phase | solid (at STP) melting point | 470 °C
Units

Thermodynamic properties

specific heat of formation Δ_fH° | solid | -1.364 kJ/g molar heat of formation Δ_fH° | solid | -451.9 kJ/mol (at STP)
Chemical identifiers
([O-])[O-].[N+](=O)([O-])[O-].[Pb+2] InChI identifier | InChI=1/2NO3.Pb/c2*2-1(3)4;/q2*-1;+2 RTECS number | OG2100000 MDL number | MFCD00011153](../image_source/3cddc3e9e405e11544537b34168a36ef.png)
CAS number | 10099-74-8 PubChem CID number | 24924 PubChem SID number | 24852183 SMILES identifier | [N+](=O)([O-])[O-].[N+](=O)([O-])[O-].[Pb+2] InChI identifier | InChI=1/2NO3.Pb/c2*2-1(3)4;/q2*-1;+2 RTECS number | OG2100000 MDL number | MFCD00011153
Toxicity properties

odor | odorless
Ion equivalents

Pb^(2+) (lead(II) cation) | 1 (NO_3)^- (nitrate anion) | 2