Input interpretation

uranium | van der Waals radius
Result
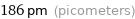
186 pm (picometers)
Periodic table location
Periodic table location
Atomic radii properties

atomic radius (empirical) | 175 pm covalent radius | 196 pm van der Waals radius | 186 pm
Units

Unit conversions
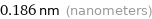
0.186 nm (nanometers)
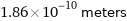
1.86×10^-10 meters
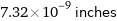
7.32×10^-9 inches
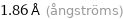
1.86 Å (ångströms)
Comparisons as atomic radius
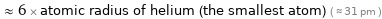
≈ 6 × atomic radius of helium (the smallest atom) ( ≈ 31 pm )
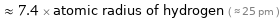
≈ 7.4 × atomic radius of hydrogen ( ≈ 25 pm )